Bleomycin- induced flagellate dermatitis: two cases report
Nouama Bouanani 1, Madiha EL Jazouly2
1, Madiha EL Jazouly2
1Department of Haematology, International Cheikh Khalifa University Hospital, Mohammed VI University of Health Sciences (UM6SS), Casablanca, Morocco; 2Department of Dermatology, International Cheikh Khalifa University Hospital, Mohammed VI University of Health Sciences (UM6SS), Casablanca, Morocco
Corresponding author: Dr. Nouama Bouanani
Submission: 28.05.2020; Acceptance: 27.07.2020
DOI: 10.7241/ourd.2020e.121
Cite this article: Bouanani N, EL Jazouly M. Bleomycin-induced fl agellate dermatitis: two cases report. Our Dermatol Online. 2020;11(e):e121.1-e121.3.
Citation tools:
Copyright information
© Our Dermatology Online 2020. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Bleomycin is an antibiotic with an antineoplastic citotoxic property. It has been used in the treatment of a variety of tumors, including Hodgkin’s lymphoma, testes carcinoma and head, malignant pleural effusions and neck squamous cell carcinoma. Bleomycin induced toxicities are more pronounced in lungs and skin due to low concentration of the metabolizing enzyme-bleomycin hydrolase in these organs. Here, we report two cases of Bleomycin-induced flagellate dermatitis in patients suffering from Hodgkin’s lymphoma after two cycles of ABVD regimen. The management of this dermatitis is symptomatic and usually does not require stopping chemotherapy.
Key words: Bleomycin; Flaagellate hyperpigmentation; Hodgkin’s lymphoma
INTRODUCTION
Bleomycin is an antibiotic with an antineoplastic citotoxic property. It has been used in the treatment of a variety of tumors, including Hodgkin’s lymphoma, testes carcinoma and head and neck squamous cell carcinoma. The side effects of bleomycin are most frequently seen in lungs and skin. Flagellate dermatitis is an uncommon cutaneous adverse reaction of bleomycin. We report two cases of flagellate dermatitis in Hodgkin’s lymphoma patients treated with bleomycin.
CASE REPORT
Case 1
A 24-year-old female Moroccan, with no prior medical history, recently diagnosed with classical Hodgkin’s disease Stage IIA, developed a linear erythematous and itchy eruption on her limbs 15 days after her first cycle of doxorubicin, bleomycin, vincristine and dacarbazine (ABVD) chemotherapy. The dermatological exam showed, linear and streaked pigmentation were seen on bilateral upper limbs, lower limbs, and over chest and back (Fig. 1). Topical corticosteroids and antihistamines were prescribed, resulting in the disappearance of the pruritus. No other skin lesions were noted. Patient achieved complete remission per positron emission tomography scan after completion of four cycles of chemotherapy, Radiotherapy IF 30 Gy.
 |
Figure 1: Linear hyperpigmented lesions on the upper limbs (patient 1). |
Case 2
A 48-year-old female with Hodgkin’s lymphoma (Stage IIIB) revealed by drenching night sweats, fever, pruritic, weight loss and cervical lymph nodes. The patient presented generalized pruritic, hyperpigmentation over neck and chest after receiving two cycles of ABVD (doxorubicin, bleomycin, vinblastine, and dacarbazine) chemotherapy regimen. Cutaneous examination revealed multiple linear erythematous streaks over the nape of neck, upper part of chest (Fig. 2), no skin biopsy was done. Symptomatic treatment with topical corticosteroids was prescribed with a good response. Patient achieved complete remission after completion of eight cycles of chemotherapy, a PET/CT scan demonstrated favorable anatomic and metabolic responses to the therapy.
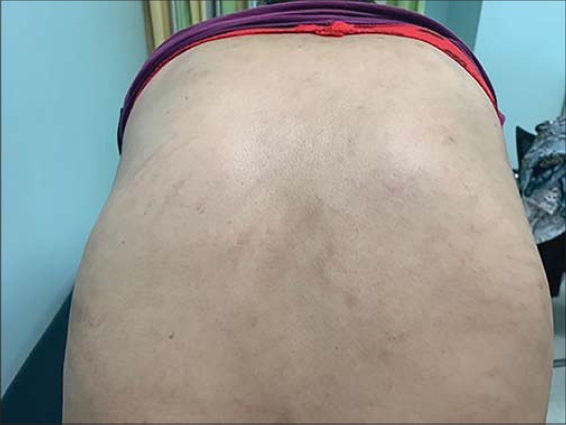 |
Figure 2: Linear erythematous lesions on the back (patient 2). |
DISCUSSION
Bleomycin is a polypeptide cytostatic derived from Streptomyces verticillus, discovered in Japan by Umezawa in 1965 [1]. It is antineoplastic antibiotic that inhibits the incorporation of thymidine into DNA, Hydrolase inactivates bleomycin in all organs except the skin and the lungs. This is considered to be the reason why most side effects of bleomycin preferably manifest in these organs [2].
Flagellate dermatitis is an uncommon adverse reaction to bleomycin, reported in 8-20% of patients under the treatment of Bleomycin [3]. It’s usually occurs after a cumulative dose of 90–285 mg of Bleomycin. But some cases have been reported with doses as low as 15 mg given parenterally [4].
The pathogenesis of bleomycin-induced pigmentation is still controversial. Several hypotheses have been proposed that included the leakage of the drug due to scratching, increased and altered melanogenesis leading to an abnormal distribution of the pigment in the horny layers [5].
Clinically, the patient can be asymptomatic or may present generalized pruritus followed by the onset of erythematous linear streaks which progress to typical flagellate hyperpigmentation. The lesions can occur anywhere over face, neck and trunk [6]. Histopathological finding is not specific. It can include hyperkeratosis, acantosis, spongyosis, basal layer degeneration, lymphohistiocytic inflammatory infiltrate in the dermis, melanophages in the papillary dermis and lymphocytic vasculities [1].
There is no specific treatment of flagellate dermatitis. No treatment modality was described in the literature. Stopping and avoiding obleomycin can lead to eventual clearance of the lesions over months to years. However, re-exposure to bleomycin may produce a new reaction [7].
Flagellate dermatitis is a benign disease whose occurrence should not put the continuation of Bleomycin therapy at risk. The management of the dermatitis is symptomatic and usually does not require stopping chemotherapy [3].
CONCLUSION
Flagellate rash also known as flagellate dermatitis is an uncommon cutaneous side effect of Bleomycin therapy. The cooperation between hematologists and dermatologists is fundamental in order to recognize this dermatitis and to make the best decisions for the patient.
ACKNOWLEDGMENTS
We thank all the staff of Mohammed VI University of Health Sciences, Casablanca, Morocco
Consent
The examination of the patient was conducted according to the Declaration of Helsinki principles.
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
REFERENCES
1. César J, Silveira GD, Cunha BM, Estrella RR. Bleomycin- induced flagellate dermatitis. An Bras Dermatol. 2006;81:83-5.
2. Grynszpan R, Niemeyer-Corbellini JP, Lopes MS, Ramos-e-Silva M. Bleomycin-induced flagellate dermatitis. BMJ Case Rep. 2013;2013:bcr2013009764.
3. Jonathan Stevens G, Mari?a Teresa Dossi C, Gianna Mun~oz M. Bleomycin-induced Flagellated dermatitis:A case report. Rev Chil Pediatr. 2018;89:257-60.
4. Narimane S, Errihani H. Dermatose flagelle?e induite par ble?omycine:un nouveau cas Clinique. Pan Afr Med J. 2016;25:86.
5. Basu D, Podder I, Das A. Bleomycin-induced flagellate hyperpigmentation:A case report with review of literature. Pigment Internat. 2016;3:40-2.
6. Agrawal C, Talwar V, Saini R, Babu P. Flagellate rash:An unusual complication of bleomycin therapy –A case report with brief review of literature. Indian J Med Paediatr Oncol. 2017;38:548-51.
7. Diao DY, Goodall J. Bleomycin-induced-flagellate dermatitis. CMAJ. 2012;184:1280.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0000-0002-7075-4030 http://orcid.org/0000-0002-7075-4030 |



Comments are closed.