Case series of a varied spectrum of drug reactions
Panchatsharam Adithyan , Bittanakurike Narasappa Raghavendra, Patra Anjan Kumar
, Bittanakurike Narasappa Raghavendra, Patra Anjan Kumar
Department of Dermatology, MVJ Medical College and Research Hospital, Bengaluru, India
Citation tools:
Copyright information
© Our Dermatology Online 2023. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Adverse drug reactions are an important cause of iatrogenic morbidity and mortality in the medical field across the globe. As newer drugs emerge into the market, newer adverse reactions are being reported. More than half of these new drugs have serious side effects. A distinction between numerous patterns of drug-induced reactions is potentially of critical importance because a delay even of several hours may make the difference between survival and death. Herein, we report five cases of severe cutaneous adverse drug reactions admitted at our dermatology ward.
Key words: Adverse drug reaction, Iatrogenic morbidity
INTRODUCTION
Adverse reactions to drugs are common in clinical practice, and reactions affecting the skin constitute a significant percentage of these reactions [1]. It is important to distinguish features of cutaneous drug reactions that help to classify the underlying mechanism, and likely prognosis, as both of these influence management decisions, some of which, necessarily, have to be taken immediately. Five cases of different morphological presentations of drug reactions will be reported here.
CASE REPORTS
Case 1
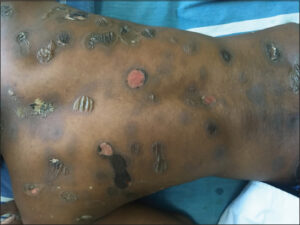
A 23-year-old male presented with complaints of fluid-filled lesions present for five days. The lesions appeared on the back, then progressed to involve the upper limbs, lower limbs, and abdomen, associated with itching and a burning sensation. He had been a known case of a seizure disorder since the age of eleven years and was on tablet phenobarbitone. He stopped tablet phenobarbitone and took sodium valproate fifteen days prior to the onset of these lesions. There was a history of five similar episodes, each lasting for 5–6 days. The fluid-filled lesions were present on the same site as in the past. A cutaneous examination revealed multiple well-defined flaccid bullae, erosions, and crusting involving the trunk, extremities, face, palms, and soles. Circumscribed areas of PIH were seen (Fig. 1). The Nikolsky sign and the bulla spread sign were negative. A diagnosis of generalized bullous fixed drug eruption (secondary to tablet sodium valproate) was established.
Case 2
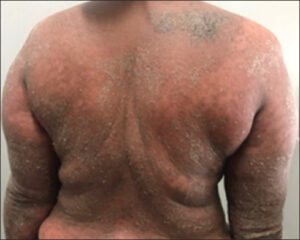
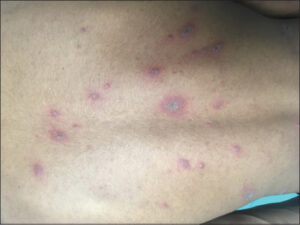
A 42-year-old female presented with complaints of itchy, red, raised lesions over the entire body present for the last two days. There was a history of fever and joint pain one week previously, for which she took tablets and injections (unknown). Two days later, red, raised lesions appeared on the trunk initially, then progressed to involve the face, both upper limbs, and lower limbs. On examination, diffuse erythema and scaling were seen involving the entire body (Fig. 2). A diagnosis of erythroderma secondary to an adverse drug reaction was established.
Case 3
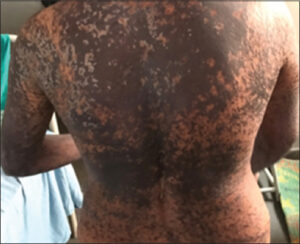
A 28-year-old female presented with complaints of red, raised lesions over the entire body associated with a severe burning sensation and itching present for the last two days. There was a history of fever five days previously, treated with tablet aceclofenac + chlorzoxazone at a local clinic. The following morning, redness and a burning sensation were initially on the forearms, then progressed to involve the entire body, including the face. On examination, multiple well-defined hyperpigmented maculopapular lesions with dusky erythema were seen over the entire body (Fig. 3). The Nikolsky sign was positive, and skin tenderness was present. Eighty percent of the body surface area was involved, with 20% epidermal detachment. On histopathology, the epidermis revealed full-thickness keratinocyte necrosis, Civatte bodies, and spongiosis. Eosinophils and lymphocytes were noted in the dermis. A diagnosis of SJS–TEN overlap syndrome secondary to aceclofenac and chlorzoxazone was established.
Case 4
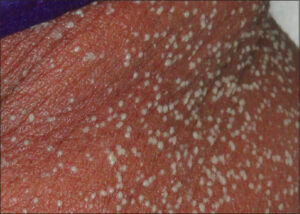
A 64-year-old female presented with a rash over the entire body present for the last four days. There was a history of high-grade fever five days back, for which the patient was prescribed tablet Augmentin 1 gm. She developed a rash within two days, which was initially on the neck and arms and gradually spread to involve the entire body. A history of an allergy to penicillin had been present since 1987, documented on records (type of reaction unknown). On examination, generalized, discrete to confluent, tiny pustules were present on an erythematous background involving the entire body sparing the palms and soles (Fig. 4). A diagnosis of acute generalized exanthematous pustulosis to the penicillin group was established.
Case 5
A 19-year-old male patient presented with complaints of watering eyes, erosions on the lips and oral cavity, and fluid-filled lesions on the trunk present for the last four days. There was a history of fever with coughing one week earlier, for which he took over-the-counter medications (Syp. dextromethorphan, oral aceclofenac, and chlorzoxazone). The following morning, the patient developed a burning sensation, watering and difficulty in opening the eyes, difficulty in swallowing, and erosions on the entire lips and oral cavity. He went to a nearby local hospital, where he was admitted and treated with injections of ceftriaxone, tablet paracetamol, and aceclofenac. The following morning, dark red lesions on the trunk, face, both upper limbs, and buttocks appeared. There was a history of similar episodes in the past. The eyes and oral cavity were involved. On examination, target lesions were seen on the trunk (Fig. 5). White patches on the tongue, conjunctival congestion, and epiphora were also seen. A provisional diagnosis of Steven–Johnson syndrome (secondary to aceclofenac and chlorzoxazone) or erythema multiforme was established. On histopathology, the epidermis revealed spongiosis, epidermal necrosis, and subepidermal bullae. The dermis showed a mixed inflammatory infiltrate composed of lymphocytes, neutrophils, histiocytes, and some eosinophils. A final diagnosis of Steven–Johnson syndrome was reached.
DISCUSSION
Drug reactions are unintended reactions that occur following the administration of drugs that are not characteristic of the desired pharmacodynamic effects of the drugs [2]. Cutaneous adverse drug reactions (CADRs) are common and account for 10–30% of all reported adverse drug reactions [3, 4], and their incidence in hospitalized patients is estimated to be 2–3%.
Drug reactions may be classified into two general categories: immunologic and non-immunologic [5]. Most adverse drug reactions are secondary to predictable, non-immunologic effects, while the residual adverse reactions are caused by unpredictable effects, some of which may be immune-mediated [6]. Only 5–10% of all adverse drug reactions are immune-mediated [7]. Immune-mediated reactions most commonly consist of either immediate or delayed immunologic mechanisms, mediated by cellular or humoral immune responses [8]. The presentation of cutaneous drug reactions may vary from localized and transient erythema to severe forms. The common CADRs are maculopapular skin rash, urticaria, fixed drug eruption (FDE), angioedema, and contact dermatitis. Serious CADRs endangering the patient’s life are Steven–Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), drug reaction with eosinophilia and systemic symptoms (DRESS), and acute generalized exanthematous pustulosis (AGEP) [9].
Once the diagnosis of CADR has been established, the most important part of the management includes the immediate discontinuation of the offending/suspected drug(s) and instituting relevant supportive and specific therapeutic measures [4]. IV corticosteroids, high dose IV IG (> 2 g/kg), appears to be a reasonable and relatively safe choice among the selective therapies currently available and previously tested. The role of steroids in the management of SJS–TEN and TEN has been controversial. Several studies have shown the possible benefit of corticosteroids. However, most of the studies have lately criticized the use of corticosteroids, stating that they not only prolong the hospital stay yet also make the patient susceptible to complications [10]. Other treatments such as cyclosporine, cyclophosphamide (100–300 mg/day), plasmapheresis, n-acetylcysteine (2 g/6 h), and TNF-a antagonists (e.g., etanercept, infliximab) have shown promising results [11].
CONCLUSION
After a cutaneous drug eruption has been diagnosed and treated, clear information must be provided to the patient regarding their drug rash. It is advisable that the patient carry a card of emergency identification that lists drug allergies and/or intolerances. The predisposition to some drug-induced eruptions may be genetic and family counseling is part of the care plan. This may be important, especially in SJS, TEN, and drug hypersensitivity syndromes.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Das NK, Martin AM. Epidemiology of cutaneous adverse drug reaction. In:Gupta LK, Martin AM (Eds). Iadvl’s textbook on cutaneous adverse drug reactions. 1st ed. Mumbai:Bhalani publishing house;2018. 9-13.
2. Roujeau J, Allanore L, Liss Y. Severe cutaneous adverse reactions to drugs (SCAR):Definitions, diagnostic criteria, genetic predisposition. Dermatol. 2009;27:2039.
3. Naldi L, Conforti A, Venegoni M, Troncon MG, Caputi A, Ghiotto E, et al. Cutaneous reactions to drugs:An analysis of spontaneous reports in four Italian regions. Br J Clin Pharmacol. 1999;48:839-46.
4. Arulmani R, Rajendran SD, Suresh B. Adverse drug reaction monitoring in a secondary care hospital in South India. Br J Clin Pharmacol. 2008;65:210-6.
5. Nayak S, Acharjya B. Adverse cutaneous drug reaction. Indian J Dermatol. 2008;53:2.
6. Joint Task Force on Practice Parameters. Executive summary of disease management of drug hypersensitivity:A practice parameter. Ann Allergy Asthma Immunol. 1999;83:665-700.
7. deShazo RD, Kemp SF. Allergic reactions to drugs and biologic agents. Jama. 1997;278:1895-906.
8. Zhao Y, Wang T, Li G, Sun S. Pharmacovigilance in China:Development and challenges. Int J Clin Pharm. 2018;40:823-31.
9. Li LF, Ma C. Epidemiological study of severe cutaneous adverse drug reactions in a city district of China. Clin Exp Dermatol. 2006;31:642-7.
10. Singh GK, Chatterjee M, Verma R. Cyclosporine in Stevens–Johnson syndrome and toxic epidermal necrolysis and retrospective comparison with systemic corticosteroid. Indian J Dermatol Venereol Leprol. 2013;79:686-92.
11. Granowitz EV, Brown RB. Antibiotic adverse reactions and drug interactions. Critical Care Clinics. 2008;24:421-42.
Notes
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
|




Comments are closed.