Lyell’s syndrome: Exceptional dermatosis in an infant
Sara El Ammari , Hanane Baybay, Oumaima Bouraqqadi, Rasha Moumna, Sara Elloudi, Meryem Soughi, Zakia Douhi, Fatima Zahra Mernissi
, Hanane Baybay, Oumaima Bouraqqadi, Rasha Moumna, Sara Elloudi, Meryem Soughi, Zakia Douhi, Fatima Zahra Mernissi
Department of Dermatology, University Hospital Hassan II, Fes, Morocco
Citation tools:
Copyright information
© Our Dermatology Online 2023. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
Sir,
Lyell’s syndrome is a serious, acute, life-threatening condition, rare in the pediatric population [1], characterized by skin detachment of more than 30% of the body surface area, with at least two mucous membranes involved [1,2]. It is considered a hypersensitivity reaction to numerous types of drugs, primarily anticonvulsants, antibiotics, and nonsteroidal anti-inflammatory drugs [2]. Infections particularly with Mycoplasma pneumoniae may also act as potential co-factors in the pediatric population [2,3]. The diagnosis is often clinical [1]. A skin biopsy is not always required in young children or if the diagnosis is obvious and encounters complete epidermal necrosis [3]. Treatment is based on the immediate discontinuation of the offending drug, prompt admission to the intensive care unit, and local care [3]. Specific treatment in children is controversial, with a lack of clinical trials, and includes systemic corticosteroids and intravenous immunoglobulin [1,3,4]. Short-term sequelae are often related to mucosal involvement dominated by synechiae [4]. Long-term complications may occur, mainly ocular, such as dry eye syndrome, genitourinary, pulmonary, and renal. Thus, long-term monitoring must be instituted [5]. Finally, raising public awareness and educating the public about the harmful effects of self-medication may help decrease the occurrence of severe cutaneous drug reactions in children [6].
Herein, we report the case of a nine-month-old infant followed for epilepsy secondary to congenital hydrocephalus. Five days before the admission, he presented a pruritic cutaneous eruption with the appearance of liquid lesions 48 hours later on the lower limbs and the face, following treatment with sodium valproate and carbamazepine with a delay of one month for the former and fifteen days for the latter. A general examination revealed an apyretic infant. A dermatological examination revealed multiple vesicular bullae and blisters on the legs, thighs, and cheeks with a positive Nikolsky sign and maculo-papular exanthema on the rest of the body (Fig. 1a). The skin area affected was initially estimated to be 25%. The ocular, oral, nasal, and genital mucosa were affected as well (Figs. 1a – 1c). After hospitalization, a biological checkup revealed creatine phosphokinase (CPK) three times the normal value, CPK mb five times the normal value, with C-reactive protein (CRP) at 33 mg/L and negative procalcitonin. A skin biopsy was not taken. Both drugs were stopped immediately and replaced by levetiracetam and clobazam. On the next day, the skin surface area was extended to 35% with a wet linen appearance (Fig. 1d). The diagnosis of Lyell’s syndrome was retained. The infant was put on rehydration, josamycin, local care, dermocorticoids for the skin lesions, a healing cream for the mucous lesions, and an antihistamine. A pharmacological investigation revealed the imputability of carbamazepine treatment. The evolution was favorable, with regression of the lesions without the appearance of short-term complications such as synechiae.
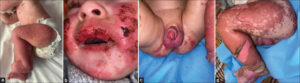 |
Figure 1: (a) Maculo-papular exanthema with multiple vesicular bullae and blisters on the legs and thighs. (b-c) Severe mucosal damage.(d) Skin detachment with a wet linen appearance. |
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Goyal S, Gupta P, Ryan CM, Kazlas M, Noviski N, Sheridan RL. Toxic epidermal necrolysis in children:Medical, surgical, and ophthalmologic considerations. J Burn Care Res. 2009;30:437-49.
2. Techasatian L, Panombualert S, Uppala R, Jetsrisuparb C. Drug-induced Stevens-Johnson syndrome and toxic epidermal necrolysis in children:20 years study in a tertiary care hospital. World J Pediatr. 2017;13:255-60.
3. Ferrandiz-Pulido C, Garcia-Patos V. A review of causes of Stevens–Johnson syndrome and toxic epidermal necrolysis in children. Arch Dis Child. 2013;98:998-1003.
4. Ramien M, Goldman JL. Pediatric SJS-TEN:Where are we now?F1000 Res. 2020;9:982
5. Lee HY, Walsh SA, Creamer D. Long-term complications of Stevens-Johnson syndrome/Toxic epidermal necrolysis:The spectrum of chronic problems in patients who survive an episode of SJS/TEN necessitates multi-disciplinary follow up. Br J Dermatol. 2017;177:924-35.
6. Tounkara TM, BaldéH, Soumah MM, Bangoura M, DianéBF, Keita M, et al. Severe cutaneous drug reactions in Guinean children:A monocentric retrospective study of 35 cases. Our Dermatol Online. 2018;9:118-22.
Notes
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0009-0001-8083-1730 http://orcid.org/0009-0001-8083-1730 http://orcid.org/0000-0003-3455-3810 http://orcid.org/0000-0003-3455-3810 http://orcid.org/0000-0002-5942-441X http://orcid.org/0000-0002-5942-441X |




Comments are closed.