Pyoderma gangrenosum mimicking a Buruli’s ulcer in an HIV-positive patient from Ivory Coast
Farah El Hadadi 1, Line Mezni1, Kaoutar Znati2, Mariame Meziane1, Nadia Ismaili1, Laila Benzekri1, Karima Senouci1
1, Line Mezni1, Kaoutar Znati2, Mariame Meziane1, Nadia Ismaili1, Laila Benzekri1, Karima Senouci1
1Department of Dermatology, Mohammed V University in Rabat, Ibn Sina University Hospital, Rabat, Morocco, 2Department of Pathology, Mohammed V University in Rabat, Ibn Sina University Hospital, Rabat, Morocco
Corresponding author: Farah El Hadadi, MD
How to cite this article: El Hadadi F, Mezni L, Znati K, Meziane M, Ismaili N, Benzekri L, Senouci K. Pyoderma gangrenosum mimicking a Buruli’s ulcer in an HIV-positive patient from Ivory Coast. Our Dermatol Online. 2022;13(2):158-160.
Submission: 30.08.2021; Acceptance: 02.02.2022
DOI: 10.7241/ourd.20222.9
Citation tools:
Copyright information
© Our Dermatology Online 2022. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
A lower limb ulcer is a disabling disease with significant functional impact and difficulties to manage. Ulcers, due to infectious agents, an inflammatory etiology, or microvascular occlusion are particularly challenging in terms of diagnosis and treatment. The management of such ulcers requires proper clinical examination as well as oriented biological and histological assessments. Herein, we report the case of a 39-year-old female from Ivory Coast with an extensive bilateral lower limb ulcer evoking a Buruli’s ulcer. Investigations, however, revealed a severe case of pyoderma gangrenosum in an HIV-positive patient. Our case report demonstrates the difficulties in the diagnosis of leg ulcers, especially in immunosuppressed patients. Confusing pyoderma gangrenosum with a Buruli’s ulcer may lead to extensive surgery or the use of long-term steroids with multiple iatrogenic complications, hence the importance of a skin biopsy and biological assessments in establishing a correct diagnosis.
Key words: Ulcer; Lower limb; Pyoderma gangrenosum; Buruli’s ulcer; HIV
INTRODUCTION
Pyoderma gangrenosum (PG) is a neutrophilic dermatosis characterized by chronic ulcers due to an abnormal immune response. Five clinical subtypes of PG exist: ulcerative, bullous, pustular, and vegetative. In the case of ulcerative PG, skin and soft tissue bacterial and non-bacterial infections, such as cutaneous tuberculosis, leishmaniasis, and Buruli’s ulcers, are common mimickers. Workup, including a biopsy, are required in all patients suspected of having pyoderma gangrenosum in order to exclude these differential diagnoses.
CASE REPORT
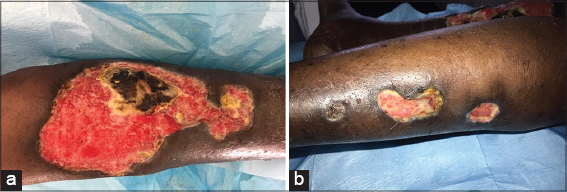
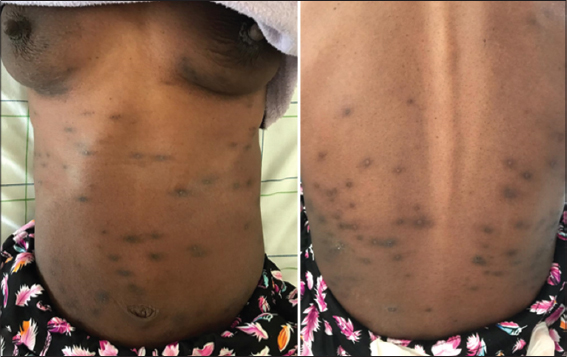
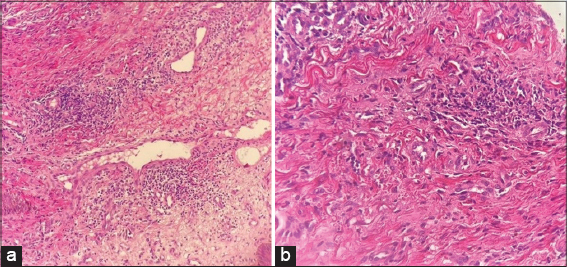
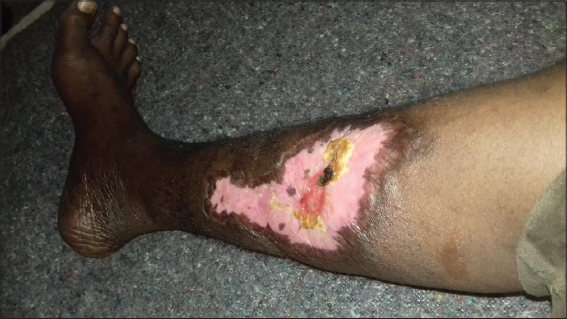
A 39-year-old female from Ivory Coast was admitted to our department for the management of multiple, non-healing, painful ulcers in the lower extremities. The skin ulcers developed six weeks earlier after the appearance of a nodule and a painful edema in the legs. There was no history of trauma. The patient took no regular medications and denied fever, chills, diarrhea, abdominal pain, or arthralgia. On admission, the vital parameters were as follows: blood pressure at 128/60 mmHg, pulse rate at 70 beats per minute, respiratory rate at 14/min, and body temperature at 37.2°C. A physical examination revealed an erythematous ulceration 15 × 10 cm in size on the anterior side of the right leg, with irregular borders a central blackish crust on the posterolateral side of the left leg, three ulcerations, with the largest 3 × 1 cm in size (Figs. 1a and 1b), a prurigo eruption on the trunk (Fig. 2), and multiple lymphadenopathies in the inguinal and cervical regions. In terms of our examination, we evoked a Buruli’s ulcer, cutaneous tuberculosis, leishmaniasis, cryptococcosis in a patient with HIV, and a pyoderma gangrenosum. Laboratory results yielded a positive HIV serology confirmed by a Western blot test. Her CD4 cell count and HIV RNA level were 92 cells/mL and 419 302 copies/mL, respectively. She had elevated C-reactive protein and an elevated erythrocyte sedimentation rate without an increase in the white cell count and a decreased hemoglobin level. She tested negative for hepatitis B, hepatitis C, and syphilis. A skin biopsy was performed on the second day of hospitalization. A histopathological examination revealed an infiltration of inflammatory cells, predominately neutrophils, extending from the dermis to the subcutis on hematoxylin and eosin staining (Figs. 3a and 3b). Special staining (Gram stain, Grocott stain, PAS stain) revealed no microorganisms. The culture of a biopsy specimen taken from the edge of the ulcer was negative for bacteria, fungi (cryptococcosis), and atypical mycobacteria (Mycobacterium ulcerans). A chest X-ray was normal, with no osteomyelitis in the lower leg X-ray. A polymerase chain reaction assay to detect Mycobacterium tuberculosis was also negative. The pathologists concluded that the histopathological findings were compatible with the diagnosis of pyoderma gangrenosum. On the day of hospitalization, the patient presented with headaches. An extensive workup for HIV (lumbar puncture, brain scan, serum electrolytes) was negative. Subsequently, an antiretroviral therapy including tenofovir disoproxil fumarate, emtricitabine, and efavirenz was started on the twentieth day of hospitalization. Local treatment consisted of vaseline tulles and hyaluronic acid cream. Within two months, the ulcers became progressively smaller and less painful and reached complete healing (Fig. 4). No other immunosuppressive therapy was added, including oral prednisone.
DISCUSSION
A Buruli’s ulcer (BU), also classified as neglected tropical disease by the WHO, is a disabling skin infection caused by Mycobacterium ulcerans. It has been reported in around 33 (mostly tropical) countries, with the greatest frequency in Africa, particularly in the West African countries of Ivory Coast, Ghana, and Benin (20 to 158 cases per 100,000) [1]. BUs preferentially affect children from two years of age without a predilection for sex. It affects females more often than males in adulthood, probably due to direct transcutaneous transmission from water. Similarly to pyoderma gangrenosum, BUs often affect the legs. However, they often begin with a painless indurated nodule or plaque, sometimes edema of a limb with local inflammatory symptoms, then appear as a profound dermoepidermal ulceration with a yellowish necrotic background. They evolve very slowly (for several months) with spontaneous scarring at the cost of irreversible sequelae (retraction, ankylosis), which differentiates them from PG (rapid evolution, painful character). The main complication of a BU is osteitis, that is why an X-ray of the limbs is mandatory. When the ulcers are multifocal and extensive, it is necessary to search for an association with an HIV infection, which is considered the main risk factor for a BU [2,3].
The clinical aspect of PG and a BU may be identical and it may be difficult to diagnose a BU. The culture of M. ulcerans on a Loewenstein–Jensen medium requires a low ambient temperature (32°C) and the growth is very slow (6–8 weeks). Acid-fast bacilli may be found in biopsy specimens (with a sensitivity of 90%) in actively ulcerating cases, yet otherwise the diagnostic yield is fairly low. Histology of a cutaneous biopsy shows necrosis of dermal collagen and subcutaneous adipose tissue with a minimal inflammatory reaction and typically the presence of more or less frequent large acid-fast bacilli. The confirmation diagnosis is based on the DNA amplification technique (PCR) of M. ulcerans.
The standard of care remains surgical excision and skin grafting, if in primary closure, is not feasible. A mistaken treatment of a BU as pyoderma gangrenosum with high-dose corticosteroids has been reported. This underlines the clinical similarity of these two entities. BUs should, therefore, be considered a differential diagnosis of pyoderma gangrenosum in any patient from an African country with lower limb ulcers [4].
CONCLUSION
To conclude, the diagnosis of leg ulcers is based on a set of arguments: the epidemiological situation of the patient completed by a biological and X-ray assessments guided by a clinical examination. An extensive ulcer of pyoderma gangrenosum may be confused with a Buruli‘s ulcer, especially in an HIV-positive patient, hence the interest in skin biopsies and bacteriological samples to avoid any invasive or extensive surgery.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Kanga JM, Kacou ED. Epidemiological aspects of Buruli ulcer in Côte d’Ivoire:Results of a national survey. Bull Soc Pathol Exotiq. 2001;94:46-51.
2. Degboe B, Sopoh GE, Alimi M, Koudoukpo C, Akpadjan F, Agbéssi N, et al. Buruli ulcer:Evaluation of its medical and surgical management at the Allada (Benin) Screening and Treatment Center, 2010-2014. Med Sante Trop. 2019;29:402-8.
3. Koudoukpo C, Atadokpede F, Salissou L, Assogba F, Akpadjan F, Degboe B, et al. [Clinical evaluation of the deadline of healing of the ulcer of Buruli hurts of diameter lower or equal to 10 centimeters in pobe (Benin)]. Our Dermatol Online. 2016;7:139-44.
4. Rouphael NG, Ayoub NM, Tomb RR. Skin ulcers misdiagnosed as pyoderma gangrenosum. N Engl J Med. 2003;348:1064-6;author reply 1064-6.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
|



Comments are closed.