A new case of scalp angiosarcoma revealed by eyelid edema
Maha Mouradi 1, Fatima Zahra Elfatoiki1, Fouzia Hali1, Farida Mernissi2, Sara Moukhlis2, Soumiya Chiheb1
1, Fatima Zahra Elfatoiki1, Fouzia Hali1, Farida Mernissi2, Sara Moukhlis2, Soumiya Chiheb1
1Department of Dermatology and Venerology,IbnRochd University Hospital, Casablanca, Morocco, 2Department of pathology, Ibn Rochd University Hospital, Casablanca, Morocco
Corresponding author: Maha Mouradi, MD
How to cite this article: Mouradi M, Elfatoiki FZ, Hali F, Mernissi F, Moukhlis S, Chiheb S. A new case of scalp angiosarcoma revealed by eyelid edema. Our Dermatol Online. 2022;13(1):77-81.
Submission: 18.01.2021; Acceptance: 07.06.2021
DOI: 10.7241/ourd.20221.19
Citation tools:
Copyright information
© Our Dermatology Online 2022. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Cutaneous angiosarcoma is a rare and highly aggressive neoplasm with poor prognosis. Owing to its variable presentation, it may be a challenging clinical and histological diagnosis. Herein, we describe a particular case of cutaneous angiosarcoma of the head and neck in a 71-year-old male with skin type V who presented with solid, progressive eyelid edema. A histological examination of skin biopsies first concluded cutaneous metastasis of an undifferentiated carcinomatous tumor. Immunostaining was essential to reach the correct diagnosis. The treatment of cutaneous angiosarcoma remains unsatisfactory. We sincerely hope that the prognosis of cutaneous angiosarcoma will be improved with the use of targeted therapies based on current genetic studies as it has been for melanoma.
Key words: Cutaneous angiosarcoma; Eyelid edema; Immunohistocytochemistry
INTRODUCTION
Angiosarcoma (AS) is a rare, highly aggressive malignant neoplasm derived from vascular endothelial cells that shows a predilection for the skin and superficial soft tissues. Three distinct clinical variants account for most cases of cutaneous angiosarcoma (cAS): idiopathic angiosarcoma of the head and neck, chronic lymphedema-associated angiosarcoma, and post-irradiation angiosarcoma. Idiopathic angiosarcoma of the head and neck is the most common variant, representing less than 0.1% of all head and neck malignancies [1]. Owing to its variable presentation, cAS may be a challenging clinical and histological diagnosis. Herein, we describe a case of cutaneous angiosarcoma of the head and neck in a 71-year-old male with skin type V who presented with solid, progressive eyelid edema.
CASE REPORT
A 71-year-old male, a diabetic and chronic tobacco user, consulted for eyelid edema associated with erosive lesions of the scalp evolving for the previous eight months. A clinical examination revealed a solid white edema of both eyelids, more marked on the left (Fig. 1), preventing the eye from opening, with some erosive lesions raising infiltrated erythematous and violaceous plaques of the scalp, overflowing on the forehead, temples, and retroauricular areas (Fig. 2). No palpable lymphadenopathy was present. There was no history of radiation, trauma, an insect bite, or preexisting skin disorders.
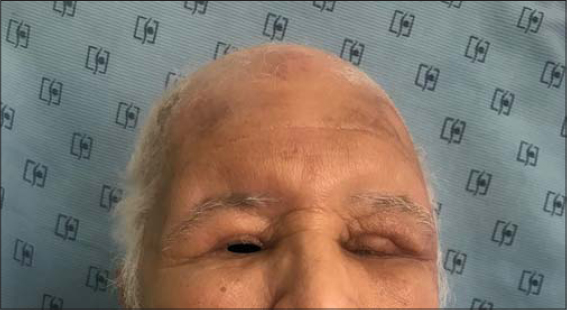 |
Figure 1: Solid white edema of both eyelids. |
 |
Figure 2: Erosive lesions raising an infiltrated erythematous and violaceous plaques of the scalp. |
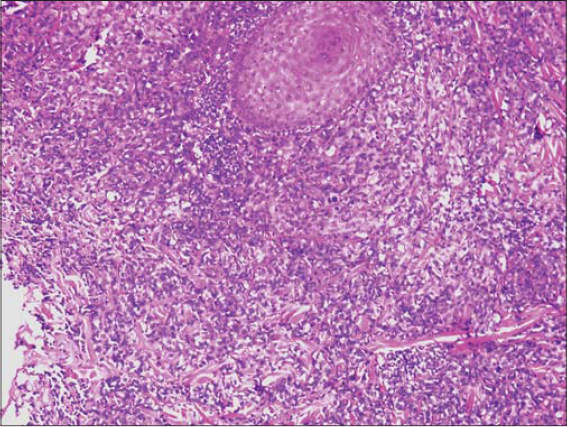
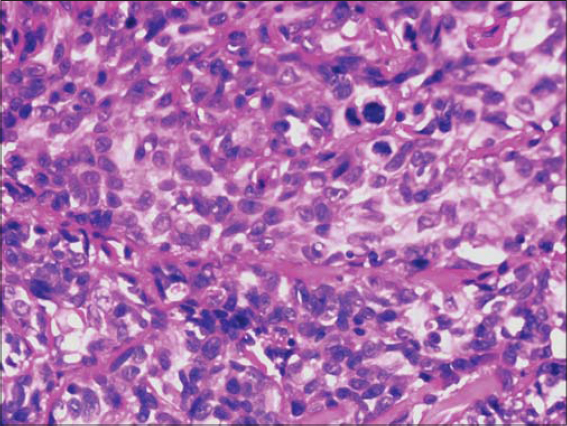
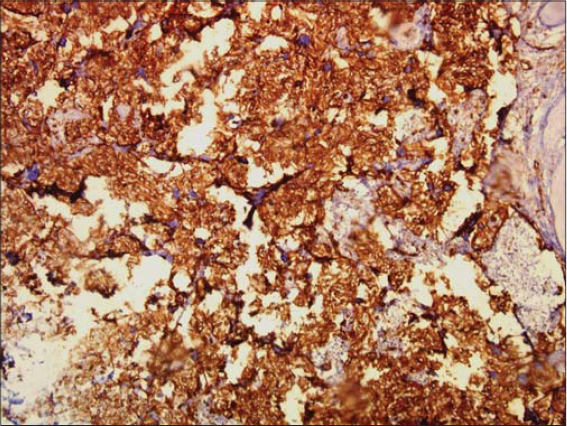
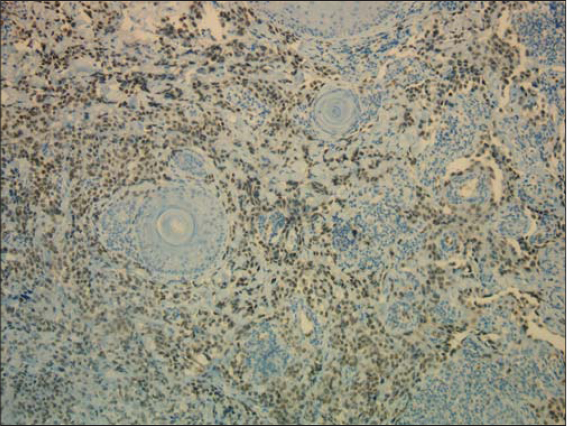
Multiple cutaneous biopsies were performed. Histology described a necrotic tumor proliferation infiltrating the dermis, made of atypical pleomorphic cells with a trabecular or lobulated appearance (Figs. 3 and 4). We first suspected cutaneous metastasis of an undifferentiated carcinomatous tumor. Immunohistochemical stains for cytokeratin markers (AE1/AE3), CK-7, CK-20, PSA, CD20 and CD3, and PS-100 were all negative, excluding a wide range of nonvascular neoplasms. Immunohistochemical stains for human herpes virus type 8 were also negative, arguing against Kaposi’s sarcoma. Having in mind the clinical progression of the purplish lesions to ulcerations and the hematoma-like lesions on the forehead, the diagnosis of cutaneous angiosarcoma was suggested, which was confirmed by diffuse and intense expression of CD31 combined with the ERG marker by the above-described tumor cells (Figs. 5 and 6). CD34 was negative. The final pathologic diagnosis was poorly differentiated cutaneous angiosarcoma. Laboratory data revealed mild leukocytosis (10,640/mL), erythrocyte sedimentation rate (29 mm/hour), and hyperglycemia 2.2g/l with a high level of HbA1c (10.7%).
A whole-body scan was performed to note mediastinal lymphadenopathies along the brachiocephalic trunk, as well as several pulmonary and hepatic nodular lesions with a metastatic appearance. Images of lysis of the lumbar vertebrae were also taken.
Chemoradiotherapy was recommended rather than extensive surgery as the patient was of an old age and the tumor was metastatic. The patient, however, declined any sort of treatment. Palliative care was provided and the patient passed away six months later at his home.
DISCUSSION
Cutaneous angiosarcoma (cAS) of the head and neck is a rare and highly aggressive neoplasm with poor prognosis, most frequently affecting the face, scalp, and neck of white elderly individuals, with males more frequently affected than females, with a ratio of 1.7:1 [2].
The usual clinical presentation of cAS is rapidly growing erythematous-to-purplish plaques or nodules with ulcerative or necrotic lesions in the progressive stages. However, cAS may mimic eczema, rosacea, hemangioma, or cellulitis, leading to delay in diagnosis [3,4]. Although case reports of eyelid and periocular lesions, as in our patient, have been reported, they are rare [5].
The etiology of cutaneous angiosarcoma is unclear. A relation to previous radiation exposure or chronic lymphedema is well established. Some cases with lesions after trauma have been described, although their significance is unknown [6]. There was no such history in our case and no known predisposing factor was identified.
Early detection by means of a biopsy is essential for a better outcome. However, because the histological features of angiosarcoma may vary both within and between cases, its histopathologic diagnosis may be challenging.
Histologically, AS is usually characterized by irregular anastomosing vascular channels with a dissecting attitude in the dermis, sometimes involving the subcutaneous fat. However, in poorly differentiated tumors, the malignant endothelial cells form continuous sheets, usually with an epithelioid morphology, with areas of hemorrhage and necrosis, which may make differentiation from anaplastic carcinoma or melanoma difficult. The presence of irregular vessels on the periphery of the tumor with small areas of hemorrhage and erythrocytes in vascular lumens may serve as diagnostic clues for vascular origin [7]. This was the case in our patient, with a poorly differentiated necrotic tumor, for which immunostaining made it possible to reach the correct diagnosis.
For this reason, we emphasize the critical importance of performing a fairly large panel of vascular markers, as some may turn negative. The cell surface markers CD31 and CD34 are the most widely employed clinically to support a diagnosis of angiosarcoma. However, these are widely expressed by other cell lineages. CD34 may be expressed in hematopoietic and fibrohistiocytic cells. CD31 is more specific, but may also be expressed in macrophages, histiocytes, and plasma cells, leading to possible diagnostic pitfalls [8].
The erythroblast transformation-specific related gene (ERG), a proto-oncogene member of the erythroblast transformation-specific transcription factor family, is thought to be highly sensitive for cAS. Associated with CD31, the ERG may corroborate a diagnosis of angiosarcoma [9]. Less frequently used vascular markers include von Willebrand factor (VWF), BNH9, factor VIII-related antigen, PROX-1, and Ulex europaeus agglutinin 1 (UEA-1).
Prognosis in cAS is poor, with five-year survival rates estimated between 12% and 20% [10]. Several factors, including the male sex, an age over fifty, a history of smoking, cardiovascular comorbidities, a location on the scalp, a tumor size of over 5 cm, the presence of satellites at the time of diagnosis, and treatment without adjuvant chemotherapy may serve as predictors for a poor prognosis [11,12].
Currently, a combination of surgery and radiation is the mainstay in the treatment of cAS. A study performed at the University of Texas MD Anderson Cancer Center demonstrated that patients who undergo combination therapy have a statistically greater overall survival rate when compared with those who undergo radiation or surgery alone [11]. However, because of delay in diagnosis, a third of the patients present with a metastatic or diffuse non-resectable tumor, as the case of our patient [13]. These patients require systemic treatments. Doxorubicin-based regimens were considered the gold standard of the treatment of soft tissue sarcomas.
Therapy with taxanes (paclitaxel) has shown better efficacy than doxorubicin with, a median progression-free survival rate of 4–5 months [14].
Successful management of infantile hemangiomas with oral and topical beta blockers led to considering propranolol for AS. Adding propranolol to a chemotherapy regimen has shown a promising response in several case reports [15,16].
The use of chemotherapy may be limited due to an advanced age or associated comorbidities. Targeted therapies, such as tyrosine kinase inhibitors (pazopanib, sorafenib, axitinib) are interesting alternatives in such cases. Bevacizumab, a VEGFR inhibitor, was reported to be an effective treatment option for AS [17]. Combinations of bevacizumab and paclitaxel are under investigation.
To date, there is no immunotherapy approved for cAS. One case report described an interesting response to anti-PD-1 treatment in a patient with angiosarcoma, although the patient experienced drug-induced hepatitis, which necessitated systemic corticosteroid treatment [18]. We sincerely hope that the treatment of cAS will be improved in the future, as it has been for melanoma.
CONCLUSION
This is a rare example of a new and challenging diagnosis of cutaneous angiosarcoma presenting as a solid, progressive eyelid edema. The case highlights the diversity of clinical manifestations associated with this type of neoplasm and the value of immunostaining in confirming a diagnosis. However, the prognosis in our case was dismal and a diagnosis at an earlier stage may have possibly given us more options for management and probably a better outcome in the end.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Maddox JC, Evans HL. Angiosarcoma of skin and soft tissue:A study of forty-four cases. Cancer. 1981;48:1907-21.
2. Dettenborn T, Wermker K, Schulze HJ, Klein M, Schwipper V, Hallermann C. Prognostic features in angiosarcoma of the head and neck:A retrospective monocenter study. J Craniomaxillofac Surg. 2014;42:1623-8.
3. Ambujam S, Audhya M, Reddy A, Roy S. Cutaneous angiosarcoma of the head, neck, and face of the elderly in type 5 skin. J Cutan Aesthet Surg. 2013;6:45-7.
4. Trinh NQ, Rashed I, Hutchens KA, Go A, Melian E, Tung R. Unusual clinical presentation of cutaneous angiosarcoma masquerading as eczema:A case report and review of the literature. Case Rep Dermatol Med. 2013;2013:906426.
5. Cabral ANF, Rocha RH, Amaral ACV, Medeiros KB, Nogueira PSE, Diniz LM. Cutaneous angiosarcoma:Report of three different and typical cases admitted in a unique dermatology clinic. An Bras Dermatol. 2017;92:235-8.
6. Mushtaq S, Sakral A, Dogra D, Bhardwaj S, Bhat A. Primary angiosarcoma of chest wall inside out:A rare presentation. Our Dermatol Online. 2017;8:306-10.
7. Paolino G, Lora V, Cota C, Panetta C, Muscardin LM, Donati P. Early angiosarcoma of the scalp:A clinicopathological pitfall. Am J Dermatopathol. 2016;38:690-4.
8. Aouali S, Alouani I, Ragragui H, Zizi N, Dikhaye S. A case of epithelioid angiosarcoma in a young man with chronic lymphedema. Our Dermatol Online. 2020;11:165-7.
9. Sullivan HC, Edgar MA, Cohen C, Kovach CK, HooKim K, Reid MD. The utility of ERG, CD31 and CD34 in the cytological diagnosis of angiosarcoma:An analysis of 25 cases. J Clin Pathol. 2015;68:44-50.
10. Donghi D, Kerl K, Dummer R, Schoenewolf N, Cozzio A. Cutaneous angiosarcoma:Own experience over 13 years. Clinical features, disease course and immunohistochemical profile. J Eur Acad Dermatol Venereol. 2010;24:1230-4.
11. Guadagnolo BA, Zagars GK, Araujo D, Ravi V, Shellenberger TD, Sturgis EM. Outcomes after definitive treatment for cutaneous angiosarcoma of the face and scalp. Head Neck. 2011;33:661-7.
12. Lee BL, Chen CF, Chen PC, Lee HC, Liao WC, Perng CK, et al. Investigation of prognostic features in primary cutaneous and soft tissue angiosarcoma after surgical resection:A retrospective study. Ann Plast Surg. 2017;78(3 Suppl 2):S41-6.
13. Becquart O, Girard C, Lesage C, Guillot B. [Angiosarcoma of the head and neck treated with bevacizumab and paclitaxel]. Ann Dermatol Venereol. 2018;145:451-3.
14. Penel N, Italiano A, Ray-Coquard I, Chaigneau L, Delcambre C, Robin YM, et al. Metastatic angiosarcomas:Doxorubicin-based regimens, weekly paclitaxel and metastasectomy significantly improve the outcome. Ann Oncol. 2012;23:517-23.
15. Agulnik M, Yarber JL, Okuno SH, von Mehren M, Jovanovic BD, Brockstein BE, et al. An open-label, multicenter, phase II study of bevacizumab for the treatment of angiosarcoma and epithelioid hemangioendotheliomas. Ann Oncol. 2013;24:257-63.
16. Banavali S, Pasquier E, Andre N. Targeted therapy with propranolol and metronomic chemotherapy combination:Sustained complete response of a relapsing metastatic angiosarcoma. Ecancermedicalscience. 2015;9:499.
17. Pasquier E, AndréN, Street J, Chougule A, Rekhi B, Ghosh J, et al. Effective management of advanced angiosarcoma by the synergistic combination of propranolol and vinblastine-based metronomic chemotherapy:A bench to bedside study. EBioMedicine. 2016;6:87-95.
18. Sindhu S, Gimber LH, Cranmer L, McBride A, Kraft AS. Angiosarcoma treated successfully with anti-PD-1 therapy – A case report. J Immunother Cancer. 2017;5:58.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
|
 http://orcid.org/0000-0002-9475-2785 http://orcid.org/0000-0002-9475-2785 |



Comments are closed.