Mucous membrane pemphigoid (cicatricial pemphigoid) with autoreactivity to salivary gland neurovascular bundles
Ana Maria Abreu Velez 1, Yuliet Alexandra Upegui-Zapata2, Michael S. Howard1
1, Yuliet Alexandra Upegui-Zapata2, Michael S. Howard1
1Georgia Dermatopathology Associates, Atlanta, Georgia, USA, 2PECET, School of Medicine, University of Antioquia, Colombia, South America
Corresponding author: Ana Maria Abreu Velez, M.D., Ph.D.
How to cite this article: Abreu Velez AM, Upegui-Zapata YA, Howard MS. Mucous membrane pemphigoid (cicatricial pemphigoid) with autoreactivity to salivary gland neurovascular bundles. Our Dermatol Online. 2021;12(3):288-290.
Submission: 02.10.2020; Acceptance: 26.12.2020
DOI: 10.7241/ourd.20213.13
Citation tools:
Copyright information
© Our Dermatology Online 2021. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Mucous membrane pemphigoid (MMP) (historically called cicatricial pemphigoid), is a rare, chronic autoimmune disease with subepithelial blisters. The blisters predominantly involve the mucous membranes, but also may affect the skin; the primary autoantigens are directed against the basement membrane zone (BMZ). A 75-year-old female presented to her dermatologist for the presence of blisters in her mouth and severe orodynia. Lesional skin biopsies were taken for hematoxylin and eosin (H&E), direct immunofluorescence (DIF) and immunohistochemistry (IHC) staining. The H&E review revealed a subepidermal blister with predominantly lymphocytes in the blister lumen, and also surrounding dermal neurovascular bundles and salivary glands. Splitting of the salivary ducts was observed. The DIF and IHC revealed the presence of polyclonal deposits at the BMZ of both the dermal/epidermal junction and salivary glands. HLA DP, DQ, DR antigen was positive in the vessels feeding the salivary glands. We further demonstrate simultaneous reactivity to other oral structures. The overall autoreactivity observed may explain the clinical scarring and compromise of salivary gland function often seen in MMP.
Key words: Mucous membrane pemphigoid; Salivary glands; Neurovascular bundles; Mesenchymal-endothelial cell junctions
Abbreviations: Mucous membrane pemphigoid (MMP); Hematoxylin and eosin (H&E); immunohistochemistry (IHC); Direct immunofluorescence (DIF); Basement membrane zone (BMZ); fluorescein isothiocyanate(FITC); 4’,6-diamidino-2-phenylindole(DAPI); Human leucocyte antigen (HLA)
INTRODUCTION
Mucous membrane pemphigoid (MMP) represents a group of autoimmune blistering diseases affecting primarily the mucous membranes, and occasionally the skin [1–4]. For decades, the disease was titled cicatricial pemphigoid, including membranous lesions with or without skin lesions. MMP usually occurs after the fifth decade of life and produces scars due to involvement of the basement membrane zone (BMZ). The oral mucosa is affected in more than 90% of cases, but other mucosal areas can be affected. Ocular lesions may result in blindness and oral lesions may cause severe airway obstruction. Though oral and ocular mucosae can both be affected in one patient, patients with only oral involvement tend to have a benign outcome [1–4]. A diagnosis requires clinical history, an H&E lesional biopsy and direct immunofluorescence (DIF) and/or immunoblotting studies to demonstrate a linear deposition of immunoglobulins (IgG and/or IgA, and or complement component 3c (C3c), at the BMZ. The target antigens vary; subsets of patients affected exclusively by oral and ocular mucosal disease have autoantibodies against a-6 and b-4 integrins, respectively [1–4].
CASE REPORT
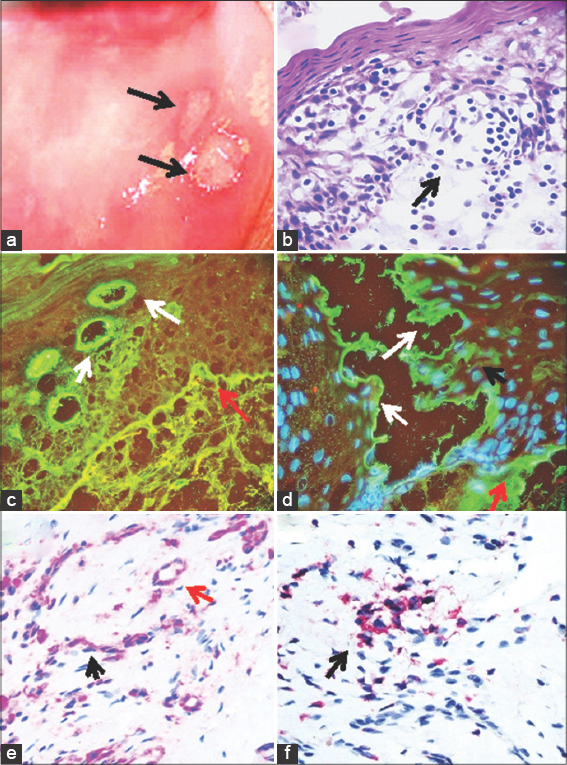
A 75-year-old female presented to her dermatologist for the presence of blisters in her mouth and severe orodynia. The physical exam revealed eroded blisters in the mouth with erythema and edema (Fig. 1a). Skin biopsies for hematoxylin and eosin (H&E) staining, for IHC and DIF were obtained. Our IHC and DIF processing were performed as previously described [5]. A diagnosis of MMP was reached, and the patient received systemic treatment with corticosteroids and mycophenolate, resulting in lesional improvement. The patient was later sent for a multidisciplinary consultation with a dentist, ophthalmologist, otolaryngologist, gynecologist, and gastroenterologist. For our DIF, we classified the findings as negative (-), weakly positive (+), positive (+++) and strongly positive (++++). IHC double staining was performed using a Leica Bond Max platform autostainer. Specifically, for red staining we utilized a bond polymer refined red detection DS9390, an alkaline phosphatase linker polymer and fast red chromogen. For brown staining, we utilized bond polymer refined detection DS9800, a horseradish peroxidase linker polymer and DAB chromogen, all from Leica (Buffalo Grove, Illinois, USA). Positive and negative controls were consistently run. We utilized antibodies against HLA-DP, DQ, DR antigen, clone CR3/43, and CD45 clones 2B11 + PD7/26, both from Dako-Agilent(Carpinteria, California, USA).
The H&E tissue sections revealed a subepithelial blister (Fig. 1b). Within the blister lumen predominantly lymphocytes were present, with occasional neutrophils and eosinophils. Within the dermis, a mild, superficial, perivascular infiltrate of lymphocytes, plasmacytoid cells, histiocytes, and a few eosinophils and neutrophils were identified near neurovascular bundles (Fig. 1b). Of interest, salivary duct lumina were filled with lymphocytes and some histiocytes. When cutting the biopsies with the microtome, the tissue easily separated at salivary gland lumina. Dermal scarring was not appreciated.
The DIF showed separation of the salivary ducts, creating a cavity. We noted IgG, IgM, IgD, anti-human fibrinogen, anti-human albumin, anti-kappa, and anti-lambda all positive (++++) with a linear distribution at the basement membrane zone (BMZ) of the dermal-epidermal junction, and also at the salivary gland BMZs (Figs. 1c and d). Deposits were also noted around upper dermal neurovascular bundles, and in mesenchymal/endothelial cell junctions throughout the dermis. Interestingly, multiple dendritic-like cells in the dermis were positive with C1q (++++). The neurovascular bundles in the dermis were also positive for FITC conjugated IgA (+++), and Complement/C3 was noted in a linear distribution at the BMZ (+++).
HLA-DP, DQ, DR antigen was positive via IHC in the salivary gland ducts and neurovascular bundles (Fig. 1e). IHC also demonstrated CD45 positive clusters as part of the inflammatory infiltrate under the blister, and around the salivary glands and neurovascular bundles (Fig. 1f).
DISCUSSION
MMP is a chronic autoimmune disease of unknown etiology, frequently involving the oral cavity and sometimes the pharynx, larynx, esophagus, glottis, eyes (causing symblepharon) and genital mucosa [1–4]. Women are often more affected than men (ratio 2:1). The most common autoantigens described for MMP are bullous pemphigoid antigen 2 (BPAG2; BP180), bullous pemphigoid antigen 1 (BPAG1; BP230), integrins a6 and b4, laminin 5(laminin 332/epiligrin, a3b3, d2 chain laminin), laminin 6 and type VII collagen [1–4]. It is characterized histologically by subepithelial blistering and by DIF featuring linear binding of IgG, IgA and C3 to the BMZ [1–4]. The predominance of mucosal involvement clinically distinguishes MMP from bullous pemphigoid. Consistent with our findings, other authors described three patients with cicatricial pemphigoid and positive immunofluorescence findings in the basement membrane zones of mucous glands in the pharynx, mouth, and nose [6,7]. The authors suggested that their findings were unique to cicatricial pemphigoid. In our case, the autoantibodies were present along the BMZ of the mucosal junctional zone, and the BMZs of the salivary glands and their ducts [6,7].
Here, we clearly demonstrate MMP autoantibodies to the basement membranes of salivary glands and their ducts, as well as to dermal neurovascular structures and the mesenchymal/endothelial cell junctions. These autoantibodies are present to multiple antibodies, complement, albumin and fibrinogen. Given our salivary gland findings, we suggest that their involvement could contribute to the scarring process.
Like ocular tears, saliva has a complex composition including electrolytes, immunoglobulins, proteins, enzymes, and mucins. Enzymes from the salivary glands begin to digest carbohydrates, enzymes from the stomach digest proteins, and enzymes from the exocrine glands of the pancreas digest carbohydrates, proteins, lipids, RNA, and DNA. Muscarinic acetylcholine receptors (mAChRs), including M1-M5 subtypes, are classic receptors in regulating water, ion, and solute transport in salivary glands [8].
MMP autoantibodies including IgG (97%), C3 (78%) IgA (27%), and IgM (12%) are directed against multiple antigens including bullous antigens 1 and 2, laminins 332 and 311, type VII collagen, a6 b4-integrin, and unidentified basal membrane zone antigens, highlighting a diverse molecular etiology including part of the cell junctions [9]. Elderly females are commonly affected, with a mean onset age of 50–80 years. The predominant mucosal sites involved are the oral mucosa, ocular mucosa, oropharynx, larynx, and genital region. Skin involvement is usually restricted to the regions of head, neck, and upper torso. A distinguishing feature of MMP is the scarring of the mucosa after the erosions and blisters heal.
CONCLUSION
Integrins are present in the salivary glands, particularly subunits beta 1, beta 3 and beta 4; this feature may explain our observed reactivity to the salivary glands [10]. The observed reactivity to the salivary glands may account for the frequent clinical scarring in MMP. Regarding the observed reactivity of the dermal neurovascular bundles, our experience when investigating multiple autoimmune skin diseases is that neurovascular bundles often demonstrate autoimmunity. In the current case we do not know the molecular etiology of this finding, warranting further studies.
REFERENCES
1. Carey B, Setterfield J. Mucous membrane pemphigoid and oral blistering diseases. Clin Exp Dermatol. 2019;44:732-9.
2. Buonavoglia A, Leone P, Dammacco R, Di Lernia G, Petruzzi M, Bonamonte D, et al. Pemphigus and mucous membrane pemphigoid:An update from diagnosis to therapy. Autoimmun Rev. 2019;18:349-58.
3. Wozniak K, Jakubowska B, Kalinska-Bienias A, Hashimoto T, Ishii N, Kowalewski C. Diagnosis of autoimmune subepidermal bullous diseases with mucous membrane involvement based on laser-scanning confocal microscopy. Eur J Dermatol. 2020 Sep 24. doi:10.1684/ejd.2020.3765.
4. Branisteanu DC, Stoleriu G, Branisteanu DE, Boda D, Branisteanu CI, Maranduca MA, et al. Ocular cicatricial pemphigoid (Review). Exp Ther Med. 2020;20:3379-82.
5. Abreu-Velez AM, Upegui-Zapata YA, Valencia-Yepes CA, Upegui-Quiceno E, Mesa-Herrera NR, Jiménez-Echavarria AM, et al. Membrane attack complex (C5b-9 complex or Mac), is strongly present in lesional skin from patients with endemic pemphigus foliaceus in El Bagre, Colombia. J Cutan Pathol. 2019;46:9259.
6. Schmidt E. Increasing the diagnostic sensitivity for mucous membrane pemphigoid by detection of salivary autoantibodies. Br J Dermatol. 2016;174:956-7.
7. Ali S, Kelly C, Challacombe SJ, Donaldson ANA, Dart JKG, Gleeson M. et al. Salivary IgA and IgG antibodies to bullous pemphigoid 180 noncollagenous domain 16a as diagnostic biomarkers in mucous membrane pemphigoid. Br J Dermatol. 2016;174:1022-9.
8. Cong X, Min SN, Wu LL, Cai ZG, Yu GY. [Role and mechanism of muscarinic acetylcholine receptor in the regulation of submandibular gland secretion]. Beijing Da Xue Xue Bao Yi Xue Ban. 2019;51:390-3.
9. Samiei M, Ahmadian E, Eftekhari A, Eghbal MA, Rezaie F, Vinken M. Cell junctions and oral health. EXCLI J. 2019;18:317-30.
10. Loducca SV, Mantesso A, Kapas S, Williams DM, Araújo NS, Araújo VC. Salivary gland tumours:immunoexpression of integrins beta 1, beta 3 and beta 4. J Oral Pathol Med. 2003;32:305-9.
Notes
Source of Support: Nil,
Conflict of Interest: Our work was supported by Georgia Dermatopathology Associates, Atlanta,
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0000-0002-7692-4133 http://orcid.org/0000-0002-7692-4133 http://orcid.org/0000-0002-9348-6227 http://orcid.org/0000-0002-9348-6227 http://orcid.org/0000-0003-0430-6093 http://orcid.org/0000-0003-0430-6093 |



Comments are closed.