Submandibular tumor mass revealing a primary basaloid squamous cell carcinoma of the parotid gland
Aya Khemir 1,2, Olfa Adouni1,2, Aïda Goucha1,2, Imene Zawati1,3, Ilhem Bettaieb1,2, Salma Kamoun1,2, Maha Driss1,2
1,2, Olfa Adouni1,2, Aïda Goucha1,2, Imene Zawati1,3, Ilhem Bettaieb1,2, Salma Kamoun1,2, Maha Driss1,2
1Department of Pathology, Salah Azaiez Institute, 9 April 1938 Avenue, Bab Saâdoun Tunis, Tunisia, 2University of El Manar of Tunis, Faculty of Medicine of Tunis, Tunisia, 3Faculty of Sciences of Tunis, University of El Manar of Tunis, Tunisia
Corresponding author: Aya Khemir, MD
How to cite this article: Khemir A, Adouni O, Goucha A, Zawati I, Bettaieb I, Kamoun S, Driss M. Submandibular tumor mass revealing a primary basaloid squamous cell carcinoma of the parotid gland. Our Dermatol Online. 2021;12(3):285-287.
Submission: 24.08.2020; Acceptance: 30.10.2020
DOI: 10.7241/ourd.20213.12
Citation tools:
Copyright information
© Our Dermatology Online 2021. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Primary squamous cell carcinoma of the parotid gland is a rare neoplasm characterized by a rapid growth and an aggressive clinical course. Basaloid squamous cell carcinoma is yet much rarer and has a dark prognosis. Through a new case report we’ll discuss anatomo-clinical features and differential diagnoses of this entity. An 83-year-old man presented with recent neck swelling. He had parotidectomy. Microscopic examination showed a malignant proliferation invading glandular parenchyma. Tumor cells were arranged in nests with peripheral palisading. They were polygonal and cohesive with abundant eosinophilic dyskeratotic cytoplasm. Perineural neoplastic invasion was observed. On different samples there were neither mucoid component nor intermediate cells. Immunohistochemical staining showed positivity of tumor cells for Cytokeratin 5/6 and P63 antibodies. The diagnosis can be retained only if a metastatic squamous cell carcinoma of upper aerodigestive tract is ruled out. To the best of our knowledge this is the second case reported in literature so far.
Key words: Salivary gland; Squamous cell carcinoma, Basaloid type; Case report
INTRODUCTION
Primary squamous cell carcinoma of salivary glands is a rare tumor. These tumors represent about 0.1 to 1.6% of all parotid gland tumors [1,2]. Basaloid squamous cell carcinoma (BSCC) is a high grade aggressive variant composed of two types of atypical cells; basaloid and mature squamous cells. This variant occurs mostly in the base of the tongue, hypopharynx and supra-epiglottic area of the larynx [3]. BSCC of the parotid gland is exceptional and to the best of our knowledge the present case is the second one reported in literature so far [4]. Through a new case report we will discuss clinical and pathological characteristics and differential diagnoses of this extremely rare tumor.
CASE REPORT
An 83-year-old male patient, without particular medical history outside a 30-pack-year smoking history, presented with left latero-cervical tumor mass. The patient reported general state alteration and rapid growth of the tumor. On physical examination the mass measured 5 cm in diameter. It was firm and seemed to be developed in the left parotid gland. At this level, skin was slightly glossy without bonding or ulcerations. No facial palsy was noted. Otherwise physical examination was normal. Sonography showed a hypoechogenic poorly circumscribed mass measuring 43 mm in diameter developed in the left parotid gland.
During surgery the tumor was tough infiltrating the sternocleidomastoid muscle (SCMM) and “sheathing” ipsilateral internal jugular vein and external carotid artery. Left total parotidectomy extended to the SCMM was made. Facial nerve was preserved.
Gross examination found expanded parotid gland measuring 7 cm in largest diameter with a pendant muscle flap. Cut surface showed a poorly circumscribed white grayish tumor measuring 45 mm in diameter. The tumor outcropped surgical margins.
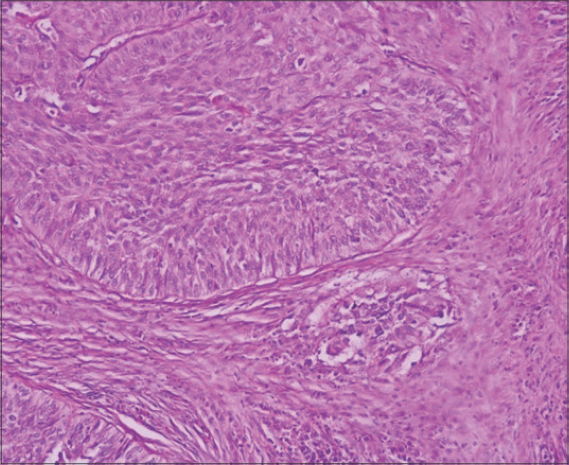
Microscopic examination revealed a carcinomatous proliferation invading glandular parenchyma made of nests and cribriform clusters with comedonecrosis (Fig. 1). Tumor cells were large, polygonal and cohesive with abundant eosinophilic cytoplasm occasionally dyskeratotic. Tumor cells had peripheral palisading (Fig. 2). Perineural neoplastic invasion was observed (Fig. 2; insert). On different samples there were neither mucoid component nor intermediate cells.
Fat and muscular tissues around the parotid gland and surgical margins were invaded indicating incomplete surgical resection. Immunostaining showed positivity of tumor cells for: Cytokeratin 5/6 and P63 antibodies. Tumor cells stained negatively for Her2neu. Therefore, the diagnosis of BSCC was retained.
The patient had adjuvant radiation therapy with positive evolution retrospectively.
DISCUSSION
Primary squamous cell carcinoma of the parotid gland is a rare tumor. Usually it’s the metastasis of a squamous cell carcinoma of upper aerodigestive tract or a local extension of a cutaneous squamous cell carcinoma of head and neck. The presence of a glandular and/or intermediate component of a high grade mucoepidermoid carcinoma, not interested in sampling, is not rare. This should be kept in mind by pathologists since it’s the most frequent parotid gland carcinoma [5].
BSCC is defined by the World Health Organization classification of head and neck tumors as an aggressive variant of high grade carcinoma that grows quickly with dark prognosis and both local and distant metastases. However, evolution depends on the tumor site (oral cavity, sinuses, hypo-pharynx) according to some cases reported in literature [6].
Medical history of radiation therapy in the area of head and neck is likely to be the main risk factor. Alterations in metaplastic cells of salivary gland ducts due to radiation therapy seem to have a key role in the pathogenesis of BSCC [5,7,8]. This is not the case of our patient who had no radiation therapy before. Smoking is not a proven risk factor of parotid gland squamous cell carcinoma in opposite to squamous cell carcinoma of upper aerodigestive tract [9]. BSCC is oftentimes positive for Human Papilloma Virus (HPV). HPV seems to be associated with a particular type of carcinoma seen usually in males in the fourth decade of life and is commonly of better prognosis with positive clinical course after radiation therapy [4,9].
The diagnosis of BSCC could be only made by pathologists. The tumor is made of basaloid-type cells with regular elongated oval nuclei. Tumor cells are arranged in lobules with peripheral palisading [4]. A cribriform pattern with comedonecrosis is commonly seen. Intracellular hyaline deposit is usually seen which mimics other salivary gland tumors. Less frequently spindle-shaped cells and pseudo-rosettes might be observed. In situ or infiltrative conventional squamous cell carcinoma may be associated [6]. Basaloid tumor cells are commonly positive for Pan-Cytokeratin (AE1/AE3, CAM5.2), heavy molecular weight cytokeratin (34BétaE12) and EMA staining. The major part of basaloid carcinoma is positive for P63 staining [3].
Main differential diagnosis is mucoepidermoid carcinoma with squamous predominant component. Broad sampling is necessary to perceive the presence of Periodic Acid-Schiff stain positive mucoid component or intermediate cells in order to rule out a high grade mucoepidermoid carcinoma. Seeking cytoplasmic intermediate filaments (tono-filaments) and intracytoplasmic desmosome-like structures on ultrastructural studies permit to make the right diagnosis. Peripheral palisading may lead to think of basal cell carcinoma which is rare in salivary glands. In fact, the presence of keratinization usually straightens the diagnosis [6]. BSCC should be distinguished from other rare tumors such as hybrid tumors, undifferentiated carcinoma and metaplastic carcinoma of the salivary gland [7,8]. Once metastatic squamous cell carcinoma of upper aerodigestive tract is ruled out the diagnosis of primary BSCC of the parotid gland may be retained. BSCC requires complete surgical excision and lymph node dissection with or without adjuvant radiation therapy [4].
In conclusion, primary squamous cell carcinoma of the parotid gland is a rare malignant neoplasm of rapid growth and aggressive clinical course. BSCC is yet much rarer and has a dark prognosis. The diagnosis of BSCC can be retained only if a metastatic squamous cell carcinoma of upper aerodigestive tract, which is more frequent, is ruled out. Despite the development of imaging and radical surgical excision techniques, the prognosis of BSCC remains dark.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Sidnoma Ouédraogo M, Korsaga/SoméN, Sosthène Ouédraogo A, Nomtondo Ouédraogo A, Gilbert Tapsoba P, Konsem T, et al. [Epidermoid carcinoma involving on chronic ulcer of the upper lip in a patient infected with HIV]. Our Dermatol Online. 2016;8:431–5.
2. BartošV. Cutaneous squamous cell carcinoma arising from the hair follicle. Our Dermatol Online. 2019;10:208–10.
3. Kulkarni AA. Primary squamous cell carcinoma of submandibular salivary gland with sialo-cutaneous fistula:a rare case report. J Clin Diagn Res . 2015;9:PD03-5.
4. Rivero A, Tang CG, Rasgon BM. Bilateral Basaloid Squamous Cell Carcinoma of the Parotid Gland:A Case Report and Review of the Literature. Perm J. 2017;21:16-042.
5. Bobati S, Patil B, Dombale V. Histopathological study of salivary gland tumors. J Oral Maxillofac Pathol. 2017;21:46.
6. El-Naggar AK, Chan JKC, Grandis JR, Takata T, Slootweg PJ, editors. WHO classification of head and neck tumours. 4th edition. Lyon:International Agency for Research on Cancer;2017. 347 p. (World Health Organization classification of tumours).
7. Seethala RR, Stenman G. Update from the 4th Edition of the World Health Organization Classification of Head and Neck Tumours:Tumors of the Salivary Gland. Head Neck Pathol. 2017 Mar;11:55–67.
8. Bansal AK, Bindal R, Kapoor C, Vaidya S, Singh HP. Current concepts in diagnosis of unusual salivary gland tumors. Dent Res J (Isfahan). 2012;9(Suppl 1):S9–19.
9. Panchbhai A. Primary squamous cell carcinoma of salivary gland:Report of a rare case. J Can Res Ther. 2015;11:664.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
|




Comments are closed.