Collision tumor: Merkel cell carcinoma and sebaceous carcinoma overlying Bowen’s disease
Ruchika Rupchandan i1, Tijjani Umar2, Stephen Walsh2
i1, Tijjani Umar2, Stephen Walsh2
1Oral and Maxillofacial Department, Western Sussex NHS Hospital Trust, UK, 2Histopathology Department, Western Sussex NHS Hospital Trust, UK, 3Oral and maxillofacial Department, Western Sussex NHS Hospital Trust, UK
Corresponding author: Ruchika Rupchandani, MD
How to cite this article: Rupchandani R, Umar T, Walsh S. Collision tumor: Merkel cell carcinoma and sebaceous carcinoma overlying Bowen’s disease. Our Dermatol Online. 2021;12(3):277-279.
Submission: 08.12.2020; Acceptance: 27.02.2021
DOI: 10.7241/ourd.20213.10
Citation tools:
Copyright information
© Our Dermatology Online 2021. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Collision tumors are the rare lesions in which two or more histopathologically distinct tumors coexist with distinct morphological features. Herein, we present the case of an 89-year-old male under clinical surveillance for multiple previous head and neck lesions. The patient presented himself with a forearm lesion, which was excised and proven by histopathology to be a collision tumor: Merkel cell carcinoma and sebaceous carcinoma were overlying Bowen’s disease. Although the patient was treated with a further wide local excision, the lesion reappeared and, despite his treatment with radiotherapy, the patient died from complications. Most combinations of collision tumors involve a basal cell carcinoma (BCC) and a melanocytic nevus. A similar case has not yet been described in the literature. The etiology of collision tumors remains unknown.
Key words: Collision tumor; Sebaceous carcinoma; Merkel cell carcinoma; Bowen’s disease
INTRODUCTION
Collision tumors are rare in the dermatology literature and refer to lesions in which two or more histopathologically distinct tumors coexist with distinct morphological features [1], including Merkel cell carcinoma and sebaceous carcinoma overlying Bowen’s disease.
Merkel cell carcinoma represents a rare neuroendocrine tumor with an incidence rate of 0.7 cases per 100,000 in the U.S. [2]. These carcinomas are associated with a high rate of local recurrence, around 21% [3], and often develop metastatic disease. On the first presentation, 26% and 8% show nodal and distant disease, respectively [4].
Sebaceous carcinomas are likewise rare tumors, with a predominance toward males (0.32 per 100,000) when compared to females (0.16 per 100, 000) [5]. These most commonly occur in the upper eyelid [6] and are tumors are associated with a 16% recurrence rate and a 20% metastatic rate [7].
Bowen’s disease, also known as carcinoma in situ, is slow-growing and responds favorably to local treatment modalities. If untreated, the lesions have the potential to transform into invasive squamous cell carcinoma, in approx. 3–5% of cases [8].
CASE REPORT
An 89-year-old male had been under clinical surveillance for six years for multiple head and neck lesions—squamous cell carcinoma (SCC) and actinic keratoses—which were fully excised. Comorbidities included osteoarthritis, glaucoma, Klinefelter syndrome, previous DVT, and paroxysmal atrial fibrillation.
During treatment for another facial SCC, the patient had a rapidly growing lesion on the right forearm, which was simultaneously excised under local anesthetic.
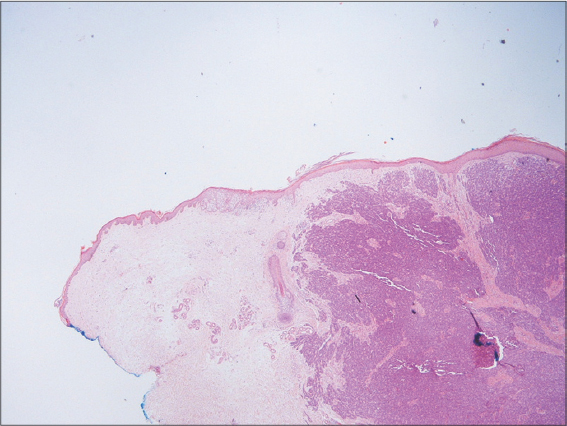
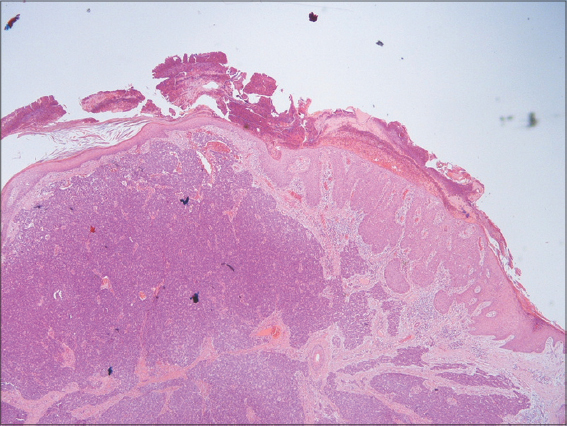
Histology of the lesion on the forearm revealed a collision tumor, involving a Merkel cell carcinoma and a sebaceous carcinoma overlying Bowen’s disease, which was completely excised.
The overlying epidermis showed proliferative squamous cell carcinoma in situ (Bowen’s disease), which was fully excised (Figs. 1 and 2).
The case was discussed in a skin multidisciplinary team (MDT) meeting after the excision. The collision tumor on the forearm had to undergo a further wide local excision with 20 mm margins of the collision tumor on the forearm and five-year clinical follow-up were recommended. CT staging was organized.
Two weeks later, the lesion was widely excised with a 2 cm margin. Histology from this wide excision revealed residual islands of Merkel cell carcinoma with no evidence of sebaceous carcinoma or Bowen’s disease. A small nodule of Merkel cell carcinoma was still abutting the peripheral resection margin. The deep resection margin was clear and another local resection was advised.
Following the re-excision, a further MDT discussion recommended radiotherapy of the local region. On the next treatment appointment, the patient had a local recurrence of the tumor, 4 × 3 cm in size at the distal end of the forearm wound.
A CT scan revealed that the tumor had spread locally to the axilla with extensive lymphadenopathy in the right subpectoral, right axillary, and right lateral chest wall lesions, but also extending up into the lower right neck. No definite lung, liver, or adrenal metastases were evident on imaging.
The patient underwent a ten-day course of palliative radiotherapy, which finished prematurely due to the COVID-19 pandemic. Then, the patient felt increasingly unwell and passed away with complications of the disease and sepsis 43 days later.
DISCUSSION
Cutaneous collision tumors are rare. A PubMed search reveals approx. 38 references of case series and reports describing 40 cutaneous collision tumors and 3 retrospective studies describing a total of 234 lesions [9,10]. Most combinations involve basal cell carcinomas (BCCs) and melanocytic nevi or melanocytic nevi and seborrheic keratoses [11]. Among the case reports and case series, only three describe collision tumors with more than two tumors within the same lesion.
Merkel cell carcinoma has previously been described with squamous cell carcinoma [12], lentigo maligna [13], and basal cell carcinoma [14]. A collision tumor involving Merkel cell carcinoma and sebaceous carcinoma has only been described in one other report: a report from 2009 on a 61-year-old Japanese male with a lesion on the eyelid [15].
As per the diagnosis and treatment of Merkel cell carcinoma, the European consensus-based interdisciplinary guidelines claim that “there is no formal evaluation of excision margins in the literature.” However, the European Association of Dermato-Oncology (EADO) and the European Organization for Research and Treatment of Cancer (EORTC) recommend a 1-to-2 cm excision margin [16]. Additionally, only in patients with clinical evidence of regional lymph node involvement should a lymph node biopsy be conducted. The report recommends a follow-up every four months during the first three years, then every six months for up to five years. However, this article describes the management of patients with metastasis as well as local disease. Merkel cell carcinoma is thought to be caused by UV light and Merkel cell polyomavirus is also implicated [17].
In the case of sebaceous carcinoma without orbital involvement, the recommended surgical therapy typically involves the excision of the visible tumor plus 5–6 mm of the surrounding healthy tissue in all directions [18]. Follow-up is recommended for at least five years [19].
A sebaceous carcinoma may be associated with Muir–Torre syndrome (MTS) in cases with mutations in DNA mismatch repair genes, resulting in microsatellite instability [20] and visceral cancers, most commonly breast and colorectal.
The etiology of collision tumors remains unknown, potentially sharing a common carcinogen (UV light). Immunodeficiency has also been suggested to play a part, as well as embryology and common derivatives of cells.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Sirikanjanapong S, Lanson B, Amin M, Martiniuk F, Kamino H, Wang BY. Collision tumor of primary laryngeal mucosal melanoma and invasive squamous cell carcinoma with IL-17A and CD70 gene over-expression. Head and Neck Pathol. 2010;4:295-9.
2. Paulson KG, Park SY, Vandeven NA, Lachance K, Thomas H, Chapuis AG, et al. Merkel cell carcinoma:Current US incidence and projected increases based on changing demographics. J Am Acad Dermatol. 2018;78:457-63.e2.
3. Santamaria-Barria JA, Boland GM, Yeap BY, Nardi V, Dias-Santagata D, Cusack JC Jr. Merkel cell carcinoma:30-year experience from a single institution. Ann Surg Oncol. 2013;20:1365-73.
4. Abreu-Velez A, Jackson B, Velasquez Cantillo K, Rafael A, Alvarez B, Howard M. Merkel cell carcinoma versus metastatic small cell primary bronchogenic carcinoma. Our Dermatology Online. 2013;4:64-71.
5. Tripathi R, Chen Z, Li L, Bordeaux JS. Incidence and survival of sebaceous carcinoma in the United States. J Am Acad Dermatol. 2016;75:1210-5.
6. Kyllo RL, Brady KL, Hurst EA. Sebaceous carcinoma:Review of the literature. Dermatol Surg. 2015;41:1-15.
7. McGrath LA, Currie ZI, Mudhar HS, Tan JHY, Salvi SM. Management of recurrent sebaceous gland carcinoma. Eye (Lond). 2020;34:1685-92.
8. Bath-Hextall F, Matin R, Wilkinson D, Leonardi-Bee J. Interventions for cutaneous Bowen’s disease. Cochrane Database Syst Rev. 2013;2013:CD007281.
9. Chung HJ, McGuigan KL, Osley KL, Zendell K, Lee JB. Pigmented solar (actinic) keratosis:An underrecognized collision lesion. J Am Acad Dermatol. 2013;68:647-53.
10. Müller C, Pföhler C, Schiekofer C, Körner R, Vogt T. Primary cutaneous carcinosarcomas. Am J Dermatopathol. 2014;36:328-39.
11. Medeiros PM, Alves NR, Silva CC, Faria PC, Barcaui CB, Piñeiro-Maceira J Collision of malignant neoplasms of the skin:Basosquamous cell carcinoma associated with melanoma. An Bras Dermatol. 2015;90(3 Suppl 1):39-42.
12. Xu H, Sonawane S, Lazzaro G, Senseng C. Collision tumor of Merkel cell carcinoma with underlying invasive squamous cell carcinoma. Am J Clin Pathol. 2018;149:16-7.
13. Forman SB, Vidmar DA, Ferringer TC. Collision tumor composed of Merkel cell carcinoma and lentigo maligna melanoma. J Cutan Pathol. 2008;35:203-6.
14. Hobbs MM, Geers TE, Brown TS, Malone JC. Triple collision tumor comprising Merkel cell carcinoma with an unusual immunophenotype, squamous cell carcinoma in situ and basal cell carcinoma. J Cutan Pathol. 2020;47:764-7.
15. Tanahashi J, Kashima K, Daa T, Yada N, Fujiwara S, Yokoyama S. Merkel cell carcinoma co-existent with sebaceous carcinoma of the eyelid. J Cutan Pathol. 2009;36:983-6.
16. Lebbe C, Becker JC, Grob JJ, Malvehy J, Del Marmol V, Pehamberger H, et al. Diagnosis and treatment of Merkel Cell Carcinoma. European consensus-based interdisciplinary guideline. Eur J Cancer. 2015;51:2396-403.
17. DeCaprio JA. Merkel cell polyomavirus and Merkel cell carcinoma. Philos Trans R Soc Lond B Biol Sci. 2017;372:20160276.
18. Grigoryan KV, Leithauser L, Gloster HM Jr. Aggressive extraocular sebaceous carcinoma recurring after mohs micrographic surgery. Case Rep Oncol Med. 2015;2015:534176.
19. Xu Y, Li F, Jia R, Fan X. Updates on the clinical diagnosis and management of ocular sebaceous carcinoma:A brief review of the literature. Onco Targets Ther. 2018;11:3713-20.
20. John AM, Schwartz RA. Muir-Torre syndrome (MTS):An update and approach to diagnosis and management. J Am Acad Dermatol. 2016;74:558-66.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0000000259858859 http://orcid.org/0000000259858859 |



Comments are closed.