Omega-3 fatty acids and quality of life in psoriasis: An open, randomised controlled study
Mohammad Adil1, Pramod Kumar Singh2, Vijay Kumar Sonkar2, Yatendra Singh Chahar2, Pankaj Kumar3, Swetank4
1Department of Dermatology, Jawaharlal Nehru Medical College, Aligarh Muslim University (A.M.U.), Aligarh, India, 2Department of Dermatology, Venereology & Leprosy, S. N. Medical College, Agra, India, 3Department of Dermatology, Venereology & Leprosy, M.R.A. Medical College, Ambedkarnagar, India, 4Department of Dermatology, Venereology & Leprosy, M.L.B. Medical College, Jhansi, India
Corresponding author: Dr. Mohammad Adil, E-mail: dr.mohd.adil@gmail.com
Submission: 13.07.2018; Acceptance: 10.09.2018
DOI:10.7241/ourd.20192.3
ABSTRACT
Background: Omega-3 fatty acids have been reported to reduce disease severity in psoriasis in various studies. This study aims to study the effects of omega-3 fatty acid supplementation on the quality of life in psoriasis.
Methods: This open, non blinded interventional study divided patients of psoriasis with affected body surface area of less than <10% into two groups of 100 patients each. Group A was given topical paraffin with 1.8 grams of omega-3 fatty acids daily in three divided doses. Group B was advised topical paraffin application once a day. Oral antihistaminics were added as per need. Baseline patient characteristics like age, sex and severity of psoriasis by Psoriasis Area Severity Index and Dermatology Life Quality Index was assessed. The patients were followed up for 12 weeks and change in DLQI was noticed.
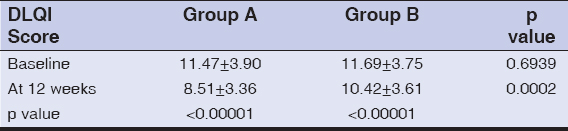
Results: Baseline DLQI in group A changed by 2.96 points from 11.47 ± 3.90 to 8.51 ± 3.36. (p <0.00001) and by 1.27 points from 11.69 ± 3.75 to 10.42 ± 3.61 in group B (p<0.00001). The change in the DLQI between the two groups was statistically significant (p=0.0002).
Conclusion: Omega-3 fatty acids appear to produce a statistically significant improvement in the quality of life in psoriasis patients.
Key words: Fish oils, Omega-3 fatty acids, Psoriasis
INTRODUCTION
Psoriasis is a chronic, immune mediated inflammatory disease characterised by well defined red scaly plaques over the extensors of body and the scalp [1]. The disease is chronic and marked by remissions and exacerbations. The clinical disease occurs due to activation of the Th1 immune response with a complex interaction of various inflammatory cells like lymphocytes, neutrophils, macrophages and dendritic cells with epidermal keratinocytes, leading to the production of various signalling molecules [2]. Psoriasis adversely impacts psychosocial well being and quality of life and may lead to self consciousness, embarrassment, social isolation and stigmatisation, leading to depression and decreased work productivity [3].
The role of diet and nutrition in the pathogenesis and management of the disease is now well established [4]. The greatest evidence in this respect has come with the use of omega-3 fatty acid supplementation [5]. Eicosapentaenoic acid (EPA) and Docosahexaenoic acid (DHA) are omega-3 fatty acids chiefly obtained from algae and marine fish. Omega-3 fatty acids lower the production of arachidonic acid derived pro inflammatory cytokines like LTB4, C4 etc. by metabolising through the same pathway as arachidonic acid and itself produces LTB5, a less potent keratinocyte stimulator and ten fold less strong chemotactic metabolite for neutrophils than LTB4 [6]. They also lead to an increased production of nitric oxide and decreased levels of Tumour necrosis factor alpha, IL-1 and other cytokines in the endothelium preventing vascular changes seen in psoriasis [7]. Altered intracellular signalling pathway, regulation of transcription factor activity and antioxidant effects have also been attributed to omega-3 fatty acids for their effect in psoriasis [8].
This study aims to find the beneficial effect of oral omega-3 fatty acids in the quality of life in patients with psoriasis.
METHODS
This prospective, open study was conducted in the department of Dermatology for a period of 2 years from March 2013 to March 2015. Ethical clearance from the institutional review board was obtained prior to the study. 100 clinically diagnosed cases of mild to moderate chronic plaque psoriasis attending the Out patient department of dermatology of the hospital (with involvement of <10% body surface area) [9] were selected and formed the study group (group A). All patients with age of 15 years to 50 years were included in the study. Patients with erythrodermic psoriasis, pustular psoriasis, palmoplantar psoriasis, pregnant and lactating women, patients with bleeding disorders, diabetes mellitus or a history of stroke or those having secondarily infected lesions were excluded from the study. Patients who were receiving any systemic therapy or phototherapy were included only after 4 weeks of washout period was passed. The same number of age and sex matched patients of chronic plaque psoriasis with <10% body surface involvement were taken as control (group B). Informed consent was given by all patients recruited for the study. Data was collected regarding the age, sex, duration of disease, age at onset and prior treatment taken. Group A was advised daily intake of 6 capsules of fish oils in three divided doses, with each capsule having 300mg of EPA and DHA (a total of 1.8 gram of omega-3 fatty acids a day). This treatment was continued for 3 months. Daily application of paraffin was advised. Antihistamines were prescribed as per need. The group B was advised daily topical paraffin therapy and antihistamines for 3 months.
Outcome of the study was measured by the change in Dermatology Life Quality Index (DLQI) score at the end of study at 12 weeks as compared to the baseline value. DLQI is a dermatology specific instrument for measuring health related quality of life index. It is a simple, 10 question validated questionnaire intended for use in adults (over 16 years of age). It is self explanatory and requires less than two minutes for completion. DLQI is calculated as the summation of the score of each question with a minimum marks of 0 and a maximum of 30 [10]. DLQI has been found to be reliable, valid, simple and easy to use [11]. Mattei et al evaluated 155 sources of randomised control trials and concluded that Psoriasis Area Severity Score (PASI) and DLQI scores are predictably correlated in patients of moderate to severe psoriasis undergoing treatment with biologicals [12].
Statistical analysis of the results regarding mean, standard deviation, percentage and tests of significance (paired and unpaired t tests and chi square tests) was done with the help of Statistical Package for Social Sciences version 16.0.
RESULTS
Both the groups in the trial consisted of 100 patients each. There were 61 males and 39 females in group A and 65 males and 35 females in group B. The mean age of patients in group A was 35.99 years with a standard deviation of 9.70 years. The group B had a mean age of 36.81 years and a standard deviation of 9.26 years. The two groups were comparable in statistical terms as far as the sex and age of the patients was concerned. The mean Psoriasis Area Severity Index (PASI) score of the two groups at baseline was also statistically not significant. The PASI score at baseline was 7.44 with a standard deviation of 2.84 for group A and 7.24 ± 2.99 for group B. After a drop out of 5 patients in group A and 7 patients in group B, 95 patients and 93 patients completed the study from the two groups respectively (Table 1). These findings have already been published in a paper by our group [13].
The Dermatology Life Quality Index for group A was 11.47 with a standard deviation of 3.90 points. This was statistically similar to the DQLI score for group B, which was 11.69 ± 3.75. The DQLI score at the end of the therapy decreased to 8.51 and with a standard deviation of 3.36 for group A and 10.42 ± 3.61 for group B. There is a significant difference between the DLQI scores of the two groups at the end of therapy at 12 weeks. The decrease in DLQI in group A was 2.96 points or 25.81%. Group B showed a decease in DLQI by 1.27 points or 10.86% (Table 2). Both the groups showed statistically significant improvements in the quality of life at the end of therapy when compared to baseline. 15 patients achieved a 75% or more reduction in PASI (PASI 75) in group A and 2 patients reached PASI 75 in group B. None of the patients experienced any adverse effect of the treatment except for fishy odor in eructation in 5 patients and slight abdominal discomfort after taking capsules in one patient. None of the patients discontinued treatment due to side effects.
DISCUSSION
Both the groups in our study were statistically comparable in terms of age, sex and severity of psoriasis assessed objectively by the Psoriasis Area Severity Index carried out by the same assessor in a patient. There was a strong male predominance in our study, with males constituting more than 60% of both the groups. Studies from the western countries show an equal predilection for both sexes [14]. This is in contrast to previous studies from Asia where a male predisposition for the disease is seen [15]. Mabuchi et al postulated that this difference might at least partly be explained by the environmental factors such as diet, smoking and alcohol, which appear to be more common in males [16]. We observed that the mean age of patients in both the groups was around 35 years of age. Our findings are in concordance with other studies showing the mean age to be in the thirties [17]. The mean PASI scores of the two groups were 7.44 and 7.24 in the two groups. It is noteworthy that patients with mild to moderate severe psoriasis (body surface area of less than 10%) [9] were included in the study. Other studies also show similar PASI scores in patients of mild to moderate psoriasis [18,19].
The DLQI in our patients was more than 11 in both the groups, signifying severe impairment of quality of life. Psoriasis patients have been shown to have a large impact on the quality of life, comparable to those with diabetes mellitus or ischemic heart disease [20]. As much as a third of all psoriasis patients suffer from pathological worry and anxiety. Around 5% have been reported to develop suicidal ideation [21]. A reliable correlation between the severity of psoriasis measured by the physician via PASI and subjectively by the patient via DLQI only exists when a large reduction of PASI score, usually by more than 75% occurs [22], problems in public and sexual interactions do not correlate with the severity of psoriasis [23]. The feeling of stigmatisation, associated co-morbidities, social withdrawal exerts an adverse effect on the educational and professional life of patients, leading to decreased quality of life [24].
The DLQI decreased significantly in both the groups. Group A showed a statistically significant greater decline in DLQI than the group B. It improved by around 25% in group A, but the decrease was a little less than 11% in group B. Several trials have evaluated the beneficial role of omega-3 fatty acids in psoriasis taking objective measures as markers of improvement, the measures being PASI, erythema, scaling, area involved etc [25]. We could find just one trial evaluating the effect of omega 3 fatty acids in psoriasis patients’ quality of life. This trial was conducted by Balbas et al, where 15 patients were advised topical tacalcitol with 640mg of omega-3 fatty acid capsules daily for 2 months. The control group was prescribed only tacalcitol for topical application. Statistically significant improvement was seen in DLQI with the test group showing an improvement of more than 6 points (78.19%) and the control group showing a 3 point improvement (53.44%) [26]. The results of our study are not as encouraging as those of Balbas et al despite using a higher dose of omega 3 fatty acids. The difference may be due to the topical agents used in the two studies. Our patients used paraffin and Balbas et al used tacalcitol, a topical vitamin D analogue.
Basra et al proposed that there should be a change in DLQI by at least 4 points to interpret it as a clinically significant change and produce a meaningful change in quality of life for the patient [27]. This means that omega 3 fatty acid supplementations failed to produce a clinically significant improvement in psoriasis in our trial, as did the group with topical emollients. The reason for this might be the low number of patients achieving PASI 75 at the end of study, indicating an incomplete clearance of the disease in most patients. Only 15 patients in group A and 2 patients in group B had achieved PASI 75 after 12 weeks. Reduction in PASI by 75% is accepted as a reliable indicator of clinical response and has been extensively used in clinical trials to gauze the efficacy of newer drugs [28]. Several authors have argued that greater reductions in PASI lead to improved quality of life as reported by the patients, with greatest reductions in quality of life parameters reported with almost complete clearance of lesions of psoriasis [29,30].
The limitations of our study included its design which was an open, non blinded trial. This predisposed to bias by both the patients as well as the assessors. The study recruited patients only with less than 10% body surface area involved. Patients of more severe psoriasis were excluded as were patients with palmoplantar psoriasis who have a small area involved but have a much greater impairment of quality of life. This study used DLQI as an assessment tool for measuring quality of life. Several newer assessment tools are now available such as Skindex, Salford Psoriasis Index and SF-36.
CONCLUSION
This study shows that omega-3 fatty acids and emollients produce a statistically significant improvement in patient reported quality of life measures in psoriasis. This improvement is only modest and may not be clinically apparent. Nevertheless, omega 3 fatty acids have a beneficial effect on other chronic diseases including ischemic heart disease, which is associated with psoriasis. These are largely safe and free of side effects. Thus, omega 3 fatty acids may be used in conjunction to other treatments in patients of psoriasis. However, more studies are needed to accurately assess the role of omega-3 fatty acids in psoriasis in improving quality of life as well as to arrive to an appropriate dosage for supplementation.
Statement of Human and Animal Rights
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Statement of Informed Consent
Informed consent was obtained from all patients for being included in the study.
REFERENCES
1. Griffiths C, Barker JN. Psoriasis. In:Burns T, Brethnach S, Cox N, Griffiths C, editors. Rook’s textbook of dermatology. 8th ed. West Sussex:Blackwell Publishing;2010. p.1-22.
2. Gudjonsson JE, Elder JT. Psoriasis. In:Goldsmith LA, Katz SI, Gilchrest BA, Paller AS, Leffel DJ, Wolff K, editors. Fitzpatrick’s dermatology in general medicine. 8th ed. New York:McGraw Hill;2012. 197-224.
3. Armstrong AW, Schupp C, Wu J, Bebo B. Quality of Life and Work Productivity Impairment among Psoriasis Patients:Findings from the National Psoriasis Foundation Survey Data 2003–2011. PLoS One. 2012;7:e52935.
4. Araujo MLD, Burgos MGAP, Moura ISCM. Nutritional influences in psoriasis. An Bras Dermatol. 2009;84:90-2.
5. Millsop JW, Bhatia BK, Debbaneh M, Koo J, Liao W. Diet and psoriasis, part III:role of nutritional supplements. J Am Acad Dermatol. 2014;71:561-9.
6. Ricketts JR, Rothe MJ, Grantkels JM. Nutrition and Psoriasis. Clin Dermatol. 2010;28:615-26.
7. Traub M, Marshall K. Psoriasis–pathophysiology, conventional and alternative approaches to treatment. Alt Med Rev. 2007;12:319-30.
8. Passi S, de Pita O, Cocchi M. Psoriasis and diet. Progress Nutrition. 2004;6:231-47.
9. Assessing a psoriasis patient. In:Voorhees AV, Feldman SR, Koo JYM, Lebwohl MG, Menter A, editors. Psoriasis and psoriatic arthritis pocket guide:treatment algorithms and management guide. 3rd ed. Portland:National Psoriasis Foundation;2009. p 9-44.
10. Finley AY, Khan GK. Dermatology Life Quality Index (DLQI) -a simple practical measure for routine clinical use. Clin Exp Dermatol. 1994;19:210-6.
11. Bronsard V, Paul C, Prey S, Puzenat E, Gourraud P-A, Aractingi S, et al. What are the best outcome measures for assessing quality of life in plaque type psoriasis? A systematic review of the literature. J Eur Acad Dermatol Venereol. 2010;24:17-22.
12. Mattei PL, Corey KC, Kimball AB. Psoriasis Area Severity Index (PASI) and the Dermatology Life Quality Index (DLQI):the correlation between disease severity and psychological burden in patients treated with biological therapies. J Eur Acad Dermatol Venereol. 2014;28:333-7.
13. Adil M, Singh PK, Maheshwari K. Clinical evaluation of omega-3 fatty acids in psoriasis. Przegl Dermatol. 2017;104:314-23.
14. Kurd SK, Gelfand JM. The prevalence of previously diagnosed and undiagnosed psoriasis in US adults:results from NHANES 2003-2004. J Am Acad Dermatol. 2009;60:218-24.
15. Dogra S, Yadav S. Psoriasis in India:prevalence and pattern. Indian J Dermatol Venereol Leprol. 2010;76:595-601.
16. Mabuchi T, Ota T, Manabe Y, Ikoma N, Ozawa A, Terui T, et al. HLA-C*12:02 is a susceptibility factor in late-onset type of psoriasis in Japanese. J Dermatol. 2014;41:697-704.
17. Puri N, Mahajan BB, Sandhu SK. Clinical evaluation of different therapeutic modalities in psoriasis by PASI score. Our Dermatol Online. 2013;4:16-22.
18. Choonhakarn C, Busaracome P, Sripanidkulchai B, Sarakarn P. A prospective, randomized clinical trial comparing topical aloe vera with 0.1% triamcinolone acetonide in mild to moderate plaque psoriasis. J Ear Acad Dermatol Venereol. 2010;24:168-72.
19. Gisondi P, Tessari G, Conti A, Piaserico S, Schianchi S, Peserico A, et al. Prevalence of metabolic syndrome in patients with psoriasis:a hospital-based case–control study. Br J Dermatol. 2007;157:68-73.
20. Langley RGB, Krueger GG, Griffiths CEM. Psoriasis:epidemiology, clinical features, and quality of life. Ann Rheum Dis. 2005;64(Suppl II):ii18–23.
21. Gupta MA, Schork NJ, Gupta AK. Suicidal ideation in psoriasis. Int J Dermatol 1993;32:188-90.
22. Puig L, Thom H, Mollon P, Tian H, Ramakrishna GS. Clear or almost clear skin improves the quality of life in patients with moderate-to-severe psoriasis:a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2017;31:213-20.
23. Schmid-Ott G, Schallmayer S, Calliess IT. Quality of life in patients of psoriasis and psoriatic arthritis with a special focus on stigmatisation experience. Clin Dermatol. 2007;25:547-54.
24. Kawro T, Zalewska-Janowska A, Hawro M, Maurer M. Impact of psoriasis severity on family income and quality of life. J Eur Acad Dermatol Venereol. 2015;29:438-43.
25. Upala S, Yong WC, Theparee T, Sanguankeo A. Effect of omega-3 fatty acids on disease severity in patients with psoriasis:a systematic review. Int J Rheum Dis. 2017;20:442-50.
26. Balbás GM, Regana MS, Millet PU. Study on the use of omega-3 fatty acids as a therapeutic supplement in treatment of psoriasis. Clin Cosmet Investig Dermatol. 2011;4:73-7.
27. Basra MK, Salek MS, Camilleri L, Sturkey R, Finlay AY. Determining the Minimal Clinically Important Difference and Responsiveness of the Dermatology Life Quality Index (DLQI):Further Data. Dermatology. 2015;230:27-33.
28. Feldman SR, Kreuger GG. Psoriasis assessment tools in clinical trials. Ann Rheum Dis. 2005;64(Suppl II):ii65-8.
29. Takeshita J, Callis Duffin K, Shin DB, Krueger GG, Robertson AD, Troxel AB, et al. Patient-reported outcomes for psoriasis patients with clear versus almost clear skin in the clinical setting. J Am Acad Dermatol. 2014;71:633-41.
30. Viswanathan HN, Chau D, Milmont CE, Yang W, Erondu N, Revicki DA, et al. Total skin clearance results in improvements in health-related quality of life and reduced symptom severity among patients with moderate to severe psoriasis. J Dermatolog Treat. 2015;26:235 9.
Notes
Source of Support: Nil
Conflict of Interest: None declared.

Comments are closed.