Our Dermatol Online. 2014; 5(4): 384-387
DOI:. 10.7241/ourd.20144.96
Date of submission: 09.07.2014 / acceptance: 13.09.2014
Conflicts of interest: None
NIMESULIDE INDUCED STEVENS JOHNSON SYNDROME (SJS); MANAGED SUCCESSFULLY WITH COMBINED APPROACH OF STEROIDS, INTRAVENOUS IMMUNOGLOBULIN AND PLACENTREX GEL: A CASE REPORT
Rakesh Tilak Raj1, Baljinder Kaur2, Krishan Kumar Lochan2, Rakesh Kumar Bahl1
1Department of Dermatology, Venereology and Leprosy, Government Medical College, Patiala (Punjab), India
2Department of Paediatrics, Government Medical College and Hospital, Patiala (Punjab), India
Corresponding author: Dr Rakesh Tilak Raj e-mail: rakeshtraj2012@gmail.com
How to cite this article: Raj RT, Kaur B, Lochan KK, Bahl RK. Nimesulide induced Stevens Johnson syndrome (SJS); managed successfully with combined approach of steroids, intravenous immunoglobulin and placentrex gel: A case report. Our Dermatol Online. 2014; 5(4): 384-387.
Abstract
There is a high mortality rate in Stevens Johnson Syndrome (SJS) and it ranges between 5%-15%. At present, there is no definite consensus regarding treatment in SJS although the effectiveness of intravenous immunoglobulin’s (IVIg) and immunosuppressive like cyclosporine have generated new hopes in the lives of these patients. But the options of combination therapy of steroids, IVIg and Placentrex gel have not been fully exercised in SJS. Henceforth, we report a case of Nimesulide induced SJS; managed successfully with a combined approach without any recurrence during a 12 months follow-up.
Key words: Severe Cutaneous adverse drug reactions (SCAR); Placentrex; Corticosteroids; Immunoglobulin’s; SCORTEN Index; Naranjo’s ADR probability Scale
| What is known? · SJS is a fatal, life threatening condition of drug therapy. · Standardized guidelines regarding treatment are lacking. · Steroids have a controversial role? · Different institutions from different countries are adopting different treatment regimens. |
Introduction
Stevens-Johnson syndrome (SJS) is a rare and life threatening form of severe cutaneous adverse drug reaction (SCAR) having an unpredictable lethal course in 5-15 % of the cases [1]. The pathogenesis of SJS is unclear and there is no universally accepted definition, but the presence of mucosal involvement and percentage of total body surface area (TBSA) affected can help in classification. Limited skin detachment i.e. epidermolysis (<10% of TBSA) favors SJS whereas widespread involvement (>30%) points towards (TEN) toxic epidermal necrolysis while intermediate cases labeled as SJS/TEN overlap according to Bastuji-Garin et al. [2]. Various etiological factors have been proposed regarding SJS but drugs are the most common cause. Levi et al. confirmed that four groups of drugs were highly suspectable to cause SJS in children < 15 years of age: sulfonamide, phenobarbitone, carbamazepine and lamotrigine [3]. The incidence of SJS in western literature was reported between 1.2-6% cases per million per year and women were more frequently affected than men [4]. In the absence of appropriate literature about the management of SJS in children and there being no established therapeutic guidelines for its management, various treatment modalities such as corticosteroids, thalidomide, pentoxifylline, plasmaphereis, cyclophosphamide, Granulocyte colony stimulating factor (GCS-F), Tissue necrosing factor -alpha (TNF- α) and cyclosporine have been tried [5]. This case report highlights, nimesulide induced SJS in a child who was successfully managed with a combined approach of steroid, IVIg and topical Placentrex gel with no recurrences even after thorough follow-up of one year.
Case Report
A 12 year old girl presented to emergency department of our hospital with the complaints of fever and rash which were intermittent in nature for six days. Those were neither associated with rigor or chills nor with diurnal variations. Patient’s mother administered 100mg of oral nimesulide on the advice of private practitioner and after six hours of drug intake, the child started complaining of irritation in her eyes and erythematous rashes over the face. Next morning, the patient’s mother noticed swelling of the face and found that the rashes had become atypical, purpuric macular rash which further progressed to involve the neck, chest, back, abdomen, thighs, legs, palms and soles within 24 hours and involved >90% of the body surface area. Oral and genital mucosa was simultaneously affected. These rashes progressed in six days to become vesicular and then bullous purpuric confluents lesions with separation of epidermis over the sites of back, abdomen, thighs and legs (Figs 1A – C). There was no history of any naso-bronchial allergy, diarrhea, upper respiratory tract infection and her past history was also noncontributory regarding any other drug allergy. On examination, she looked ill, febrile and toxic. Her vitals were stable except for tachycardia (pulse>150/min). er oral lesions revealed hemorrhagic crusted lips with difficulty in opening her mouth along with ocular examination showing bilateral congested conjunctiva with matted eyelashes having mucopurulent discharge. Genital examination showed multiple erosions over her vulva with difficulty in micturation. Nikolskys sign was positive and her higher mental functions and other systemic examinations were normal.
Laboratory work up was as under
Complete blood count (CBC) showed Hb-9.4 gm/dl,TLC-10000; DLC (N-78, L-12, E-8, M-2, B-0) and Platelet count- 280,000/ cmm3. There was mild derangement in liver function tests with SGOT -89 IU/L, SGPT-65 IU/L, RBS 252 mg%, Urine sugar was 4+, 70-80 Red blood cell/high power field. The coagulation profile, Chest X-ray, renal function test, Alkaline Phosphate, Bilirubin, erythrocyte sedimentation rate (ESR), anti-nuclear antibody (ANA), serum electrolytes, serum amylase were within normal limits. Histopathology of skin was denied by the parents of the child. Tzanck smears; skin, blood, urine, fungal and wound culture were negative. Serological studies for herpes, Epstein Barr Virus (EBV), and Cytomegalo Virus (CMV) were negative. On the basis of history of drug ingestion and clinical features supported by laboratory findings; the case was diagnosed as a Nimesulide induced SJS. Patient was admitted to the intensive care unit (ICU) and all earlier medication was immediately stopped. She was evaluated by SCROTEN index at the time of admission to predict the mortality for the child, that was 3 (pulse rate >120/min, body surface involvement >30%, blood glucose levels as >14 mmol/L) which corresponded to an expected mortality of >35.8%. This case was managed with multidisciplinary approach. Fluid requirements were calculated like burns unit. She received systemic dexamethasone (4-8 mg per day) for first three days which was tapered off gradually and on 3rd day intravenous immunoglobulin’s @0.6g/kg/day (dose used was 12g per day) was administered and it continued for four days (total dose used was 48gm). The third generation cephalosporin was added to prevent any super-added infection during the course of disease. Oral and ocular lesions were managed by topical steroids locally. Epidermal wounds were treated on the pattern of burn patients with placentrex gel topically. 10U of plain insulin was administered to control blood glucose levels. On seventh day, patient opened her eyes and her eyelid adhesions were broken by eye surgeon. On 14th day, her lesions regressed, she improved and recovered uneventfully. She was discharged after a stay of 28 days in the hospital with her laboratory reports returning to normal. Regular post SJS follow-ups at the interval of two weeks were normal showing erythema on palms and soles along with hyper and hypo-pigmentation over the involved sites (Figs 2A – C). Oral re-challenge test with Nimesulide was not done on ethical grounds as the potential risk of death outweighs therapeutic benefit. There were no side effects to IVIg, steroid and placentrex gel combination in our patient at the time of discharge with a clear instruction to avoid the culprit drug and after twelve months of follow-up, millia formation were seen on lateral aspects of neck (Fig. 3) along with the persistence of hypo/ hyper pigmentation of the involved sites. No adverse effect related to IVIg was observed in our patient.
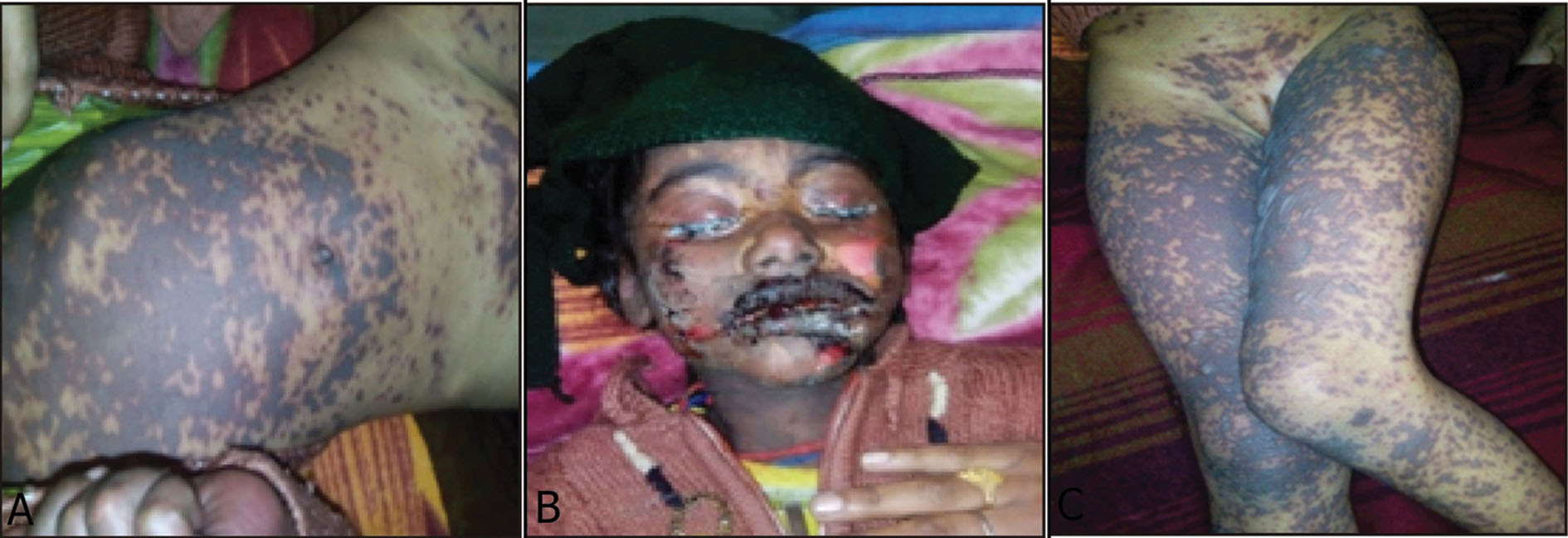 Figures 1A – C. Patient at the time of emergency showing atypical purpuric and bullous target lesions on abdomen, thighs, legs, face. neck and lips. There was haemorraghic crusting of the lips and mucopurulent discharge from her eyes.
|
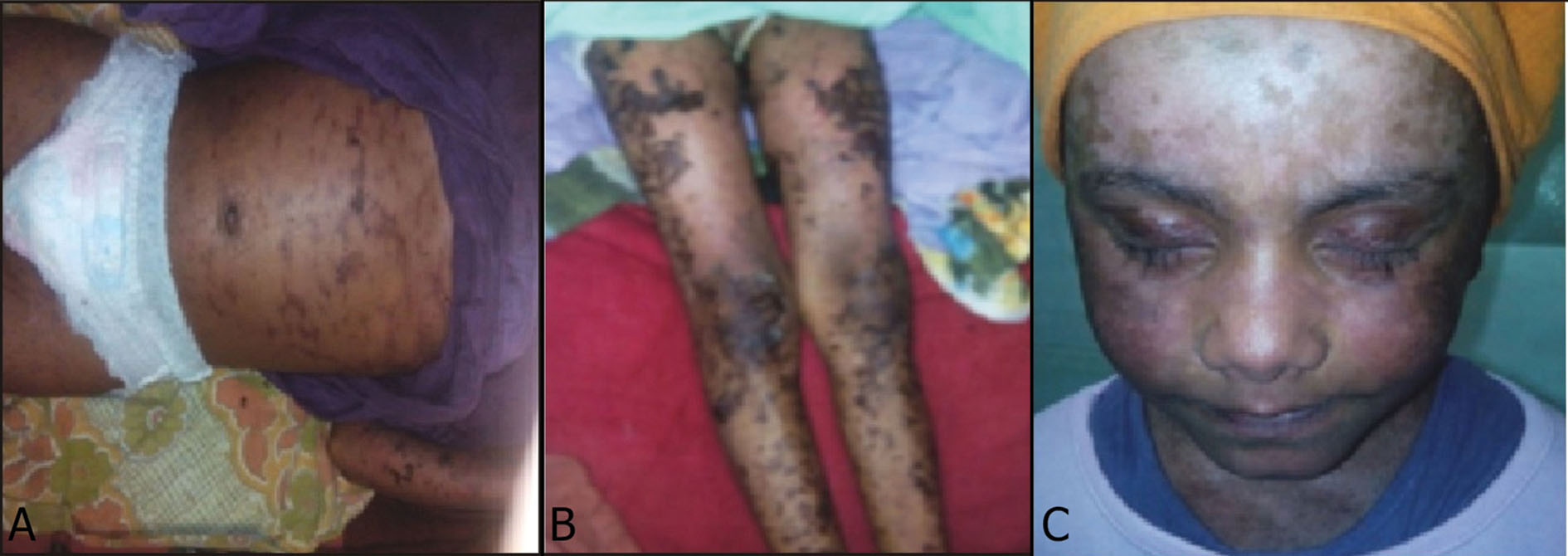 Figure 2A – C. Patient showing hyperpigmentation during recovery phase after the combined approach.
|
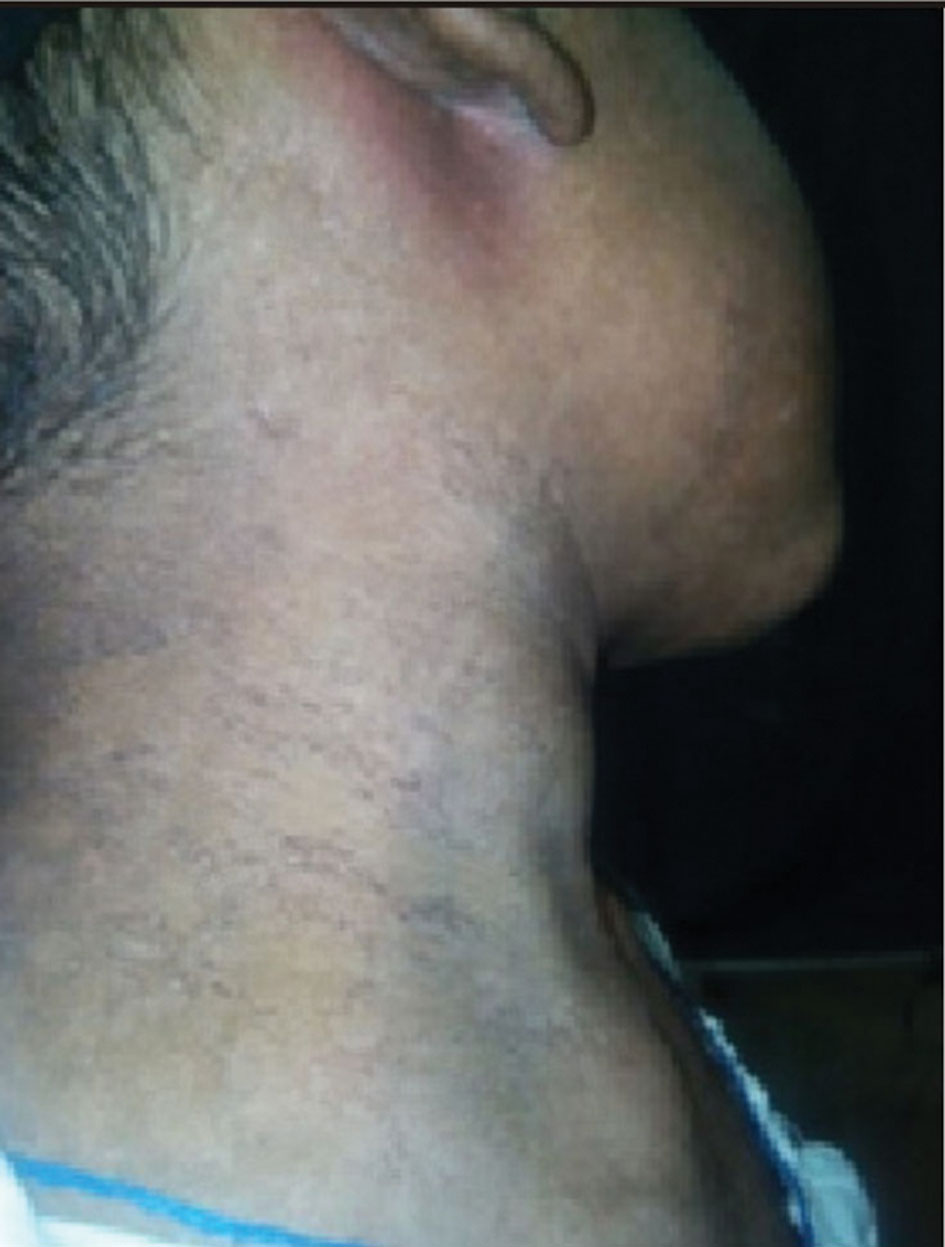 Figure 3. Patient having millia formation over her neck after 6 months of her follow up.
|
Discussion
The aetiopathogenesis of Stevens Johnson syndrome (SJS) is still unknown and drugs are commonly documented agents involved in its etiology [2]. In India, Nimesulide is easily available as an over the counter drug (OTC). Nimesulide, a selective cyclo-oxygenase inhibitor (COX-2) have been associated with fatal adverse effects due to nimesulide induced hepatotoxicity i.e. 0.1% case per one million cases treated [6] and nimesulide induced SJS has been rarely reported in the literature. Indian government restricted its use in 2011 for paediatric purposes in the age group of less than 12 years [7]. Immunological mechanisms involved in the causation of SJS are by inducing apoptosis of keratinocytes by the releasing cytokines such as IL-6, TNF- α and CD95 system through death receptors (CD 95RL/FasL, CD 95R/Fas). These receptors are a group of glycoproteins within keratinocytes which binds with Fas ligand (Fas-L) initiating apoptosis through caspases leading to DNA disassembly and cell death [8]. Diagnosis of SJS mainly relies on clinico-pathological features and its treatment consists of prompt diagnosis, discontinuation of suspected drug, appropriate symptomatic medication, fluid replacement and meticulous wound care. At present, there is no uniform strategy for managing SJS and administration of corticosteroids is considered controversial [9]. Tripathi et al. considered corticosteroids in the management of SJS in 67 patients and found them effective if given early in the disease and gradually tapered off within 72 hrs from the appearance of epidermal lesions [10]. The Rationale for the use of steroids in TEN/SJS is their role in modulation of cytotoxic T-lymphocytes releasing perforin and granzyme B in the destruction of the epithelium as they also inhibit interferon gamma mediated apoptosis[11]. IVIg is a safe and useful method of treatment in children by blocking Fas antibodies in vitro and preventing apoptosis by the formation of Fas -Fas ligand compounds [12]. The effective drug dosages of IVIg range from 0.2 to 2g/kg/day [13,14]. In this context, combination therapy seems an attractive option as they have a synergistic action targeting different pathways of apoptosis active in TEN/SJS [15]. Placentrex is a biogenic stimulator for humoral and cell mediated immunity that prevents relapse of the disease and increases body resistance. Topical human placentrex in this case has been used for its wound healing and immune-modulatory properties by suppressing interleukin-8 (an inflammatory mediator) by glucocorticoid components present in the Placentrex. [16] The aqueous extract of human placenta used as wound healer is a peptide of 7.4 kDa, its size and partial amino acid sequence indicates its similarity to human fibronectin type III. Fibronectin are adhesive mosaic glycoproteins that maintain normal cell morphology, cell migration, homeostasis, thrombosis and wound healing [17]. In this case, the diagnosis of nimesulide induced SJS was based on history, clinical examination and laboratory findings since the parents of the child denied the consent for histopathological examination. To evaluate prognosis in this case, we used SCROTEN disease severity index [18] to predict the mortality of >35.8%. Naranjo’s adverse drug reaction probability scale [19] was used to assess the causal relationship between the drug consumed by the patient and the adverse effect she developed later on. The Naranjo’s score in this case was 8 suggesting a probable temporal relationship with the drug as no other drug was taken concurrently. We use systemic corticosteroids early in the course (within 72 hours) of the disease to halt possible tissue damage from cytotoxic T-lymphocytes releasing perforin and granzyme B in the destruction of the epithelium to suppress the immune response and then added IVIg to stop Fas-mediated keratinocytes apoptosis. Placentrex was used as a biogenic stimulator for epidermal lesions to increase cell mediated immunity locally thus, acting as a wound healer. Therefore, this case report lends support to the view that IVIg is safe and effective in Nimesulide induced SJS in children and addition of low dose steroids with topical Placentrex gel resulted in enhanced healing and better survival indicating combined approach to be superior to monotherapy. However, further multicentric, randomized controlled trials are required to validate these findings.
| What is new? · Combined approach with steroid, IVIg and Placentrex may be an answer to management of SJS. · Use of Placentrex gel in place of topical steroids or topical antibiotics is more effective in healing of epidermal lesions. |
Acknowledgement
We gratefully acknowledge the efforts of postgraduate students of paediatrics department for providing round the clock care, monitoring and coordinating with other departments which helped in saving the precious life of this patient.
REFERENCES
1. Ghislain PD,Roujeau JC. Treatment of severe drug reactions: Stevens Johnson Syndrome, toxic epidermal necrolysis and hypersensitivity syndrome. Dermatol Online J. 2002;8:5.
2. Bastuji-Garin S, Rzany, Stern RS, Shear NH, Naldi L, Roujeau JC. Clinical Classification of cases of toxic epidermal necrolysis, Steven- Johnson syndrome and Erythema multiforme. Arch Dermatol. 1993;129:92-6.
3. Levi N, Bastuji- Garin S, Mockenhaupt M, Roujeau JC, Flahault A, Kelly JP, et al. Medication at risk factors for Stevens Johnson syndrome and toxic epidermal necrolysis in children: a pooled analysis. Pediatrics. 2009:123:297-304.
4. Roujeau JC1, Kelly JP, Naldi L, Rzany B, Stern RS, Anderson T, et al. Medication use and the risk of Stevens Johnson Syndrome or toxic epidermal necrolysis. N Engl J Med. 1995;333:1600-7.
5. Allanore LV, Housz Oro SI, Chosidow O, Wolkenstein P. French referral centre management of Steven Johnson syndrome/ toxic epidermal necrosis.Review article. Dermatol Sinica. 2013;31:191-5.
6. Chatterjee S, Pal J, Biswas N. Nimesulide induced hepatitis and toxic epidermal necrolysis. J Postgrad Med. 2008;54:150-1.
7. Jaykare S, Mothghare V, Pise H. Nimesulide induced Steven Johnson Syndrome: A Case report. Indian J Med Case Report. 2012;1:1-3.
8. Wehrli P, Viard I, Bullani R, Tschopp J, French LE. Death receptors in cutaneous biology and disease.Review. J Invest Dermatol. 2000;115:141-8.
9. Depew CL. Toxic epidermal necrolysis. Crit care Nurs. Clin North Am. 1991;3:255-67.
10. Tripathi A1, Ditto AM, Grammer LC, Greenberger PA, McGrath KG, Zeiss CR, et al. Corticosteroid therapy in an additional 13 cases of stevens Johnson syndrome: a total series of 67 cases. Allergy Asthma Proc. 2002;21:101-5.
11. Pereira FA, Mudgil AV, Rosmarin DM. Toxic epidermal necrolysis. J Am Acad Dermatol. 2007;56:181-200.
12. Viard I, Wehrli P, Bullani R, Schneider P, Holler N, Salomaon D, et al. Inhibition of toxic epidermo-necrolysis by blockage of CD95 with human intravenous immunoglobulin’s. Science. 1998;282:490- 3.
13. Dahl MV, Bridges AG. Intravenous immunoglobulin fighting antibodies with antibodies. J Am Acad Dermatol. 2001;45:775-83.
14. Stella M, Cassano P, Ballero D, Clemente A, Giorio G. Toxic epidermal necrolysis treated with intravenous high dose immunoglobulin: our experience. Dermatology. 2001;203:45-9.
15. Jagadeesan S, Sobhana kumari K, Sadanandan SM, Ravindran S, Divakaran MV, Skaria L, et al. Low dose intravenous immunoglobulins and steroids in toxic epidermo necrolysis. A prospective comparative open-labelled study of 38 cases. Indian J Dermatol Venereol Leprol. 2013;79:506-11.
16. Rosen T, Krikun G, Ma Y, Wang EY, Lockwood CJ, Guller S. Chronic antagonism of nuclear factor- kappa B activity in cytoblasts by dexamethsone: a potential mechanism for anti-inflammatory action of glucocorticoids in human placenta. J Clin Endocrinol Metab. 1998; 83:3647-52.
17. Chakraborty PD, Bhattacharya D. Isolation of fibronectin type III like peptide from human placental extract used as wound healer. J Chromotogr B Analyt Technol Biomed Life Sci. 2005;818:67-73.
18. Bastuji-Garin S, Fouchard N, Bertocchi M, Roujeau JC, Wolkenstein P. SCORTEN: a severity-of-illness score for toxic epidermal necrosis. J Invest Dermatol. 2000;115:149-53.
19. Naranjo CA, Busto U, Sellers BM, Sandor P, Ruiz I, Roberts EA, et al. A method for estimating the probability of adverse drug reactions. Clin Pharmacol Ther. 1981;30:239-45.
Comments are closed.