Abstract
Introduction: Pemphigus foliaceus (PF) is endemic in some South American countries, especially in Colombia and Brazil; in Brazil, it is also known as fogo selvagem (FS). We aimed to study the presence of mast cells and the expression of the mast/stem cell growth factor receptor (c-kit/CD117) in PF skin biopsies, as well as the role of IgE in the disease pathogenesis.
Methods: Forty-four skin biopsies from patients affected by endemic PF (EPF) (30 patients from El Bagre, Colombia, and 14 from the northeastern region of São Paulo State, Brazil), 48 control biopsies from Colombian and Brazilian endemic areas, and additional control biopsies from none endemic areas in Colombia and the USA non were studied. Immunohistochemistry (IHC) was performed to evaluate skin biopsies with anti-mast cell tryptase (MCT), anti-c-kit and anti-IgE antibodies. We also searched for serum IgE in 30 EPF and 30 non-atopic controls from the El Bagre region via ELISA. In our El Bagre patients and controls, we also searched for IgE in skin samples by direct immunofluorescence.
Results: In 100% of the EPF biopsies, MCT, c-kit and IgE were identified with stronger expression relative to control biopsies, especially in the inflammatory infiltrates around upper dermal blood vessels and dermal eccrine glands. IgE staining was positive along the BMZ in some EPF skin samples. The DIF results confirmed a linear deposition of IgE at the BMZ. Increased IgE serum levels were also noted in PF patients relative to controls..
Conclusions: In patients with EPF, the observed increased expression of MCT, c-kit and IgE in lesional skin, associated with higher serum IgE levels may indicate possible IgE participation in the antigenic response. .
Key words: autoimmunity; c-kit, immunoglobulin E; mast cell tryptase; endemic pemphigus; autoimmune blistering skin diseases
Abbreviations and acronyms: Endemic pemphigus foliaceus (EPF), El-Bagre endemic pemphigus foliaceus (El Bagre-EPF), direct and indirect immunofluorescence (DIF and IIF), fogo selvagem (FS), immunohistochemistry (IHC), mast cell tryptase (MCT), immunoglobulin E (IgE), mast/stem cell growth factor receptor (c-kit/CD117).
Capsule Summary:
· MCT, c-kit and IgE expression in lesional skin is higher in PF patients relative to controls.
· Serum IgE is increased in PF patients relative to controls.
· IgE may participate in the PF antigenic response.
Introduction
Endemic pemphigus foliaceus (EPF) is an autoimmune blistering skin disease presenting in well-defined geographic regions [1-7]. The roles of IgG autoantibodies and complement in lesional EPF skin has been previously documented [1-7]. Based on the fact that EPF occurs in tropical areas, and that previous authors have reported high levels of IgE in serum samples of patients affected by EPF in Brazil and Colombia [8,9], we attempted to confirm the presence of IgE in lesional skin with the purpose of confirming a potential role of IgE in EPF pathogenesis. In addition, we investigated whether mast cells and mast/stem cell growth factor receptor (c-kit/CD117) expression in lesional skin could be associated with EPF autoimmunity. Given previously documented descriptions of IgE in bullous pemphigoid [10,11], we also included skin biopsies from patients with other blistering diseases for comparison. Thus, we tested for 1) mast cell tryptase (MCT) and c-kit/CD117 expression by using immunohistochemistry (IHC), and for 2) IgE via IHC and direct immunofluorescence (DIF) in lesional skin of patients affected by EPF, and in the control biopsies.
Material and Methods
We tested 44 paraffinized skin biopsies from patients affected by EPF (30 patients from El Bagre, Colombia, and 14 from the northeastern region of São Paulo State, Brazil). All EPF patients fulfilled the established criteria for PF in Colombia [4,5] and in Brazil [2,3]. As controls, we utilized 30 skin biopsies from healthy individuals from the El Bagre endemic area (without any history of an atopic disorder). In addition, we tested 3 normal skin biopsies from Brazil, and 15 plastic surgery patients with abdominal or breast reductions from the USA or Colombian non-endemic regions. Our EPF and endemic area control biopsies were taken predominantly from areas on the chest, arms or face. A medication history was also taken, and all cases and controls were required to have had no immunosuppressant medications for at least 4 weeks. The same criteria were utilized for our serological studies. From all these patients we obtained written consents, as well as Institutional Review Board permission.
Immunohistochemistry in paraffinized skin samples
We utilized monoclonal and polyclonal antibodies from Dako (Carpinteria, California, USA). Specifically, we tested for monoclonal mouse anti-human MCT, catalogue number M7052, dilution 1:250; for polyclonal rabbit anti-human CD117/c-kit, catalogue number A4502, dilution 1:200; and for polyclonal rabbit anti-human IgE, catalogue number A0094, dilution 1:750 as previously described [13]. For our IHC testing, we utilized a dual endogenous peroxidase blockage, with the addition of an Envision dual link (to assist in chromogen adherence). We further utilized the chromogen 3,3-diaminobenzidine (DAB), and counterstained with hematoxylin. The samples were run in a Dako Autostainer Universal Staining System. Positive and negative controls were consistently performed.
Quantitative digital morphometry of staining intensity to obtain precise data on IHC parameters
The staining intensity of all antibodies was evaluated qualitatively by two independent observers, as well as via a semiquantitative approach by an automated computer image analysis system designed to quantify IHC staining in hematoxylin counterstained histologic sections. Slides were scanned with a ScanScope CS system (Aperio Technologies, Vista, California, USA), utilizing brightfield imaging at 200× and 400× magnifications. We then calculated the area of positive signal, divided by the studied area. The intensity of the staining was then classified on a scale from 0 to 4, where 0 represented negative staining and 4 the strongest staining.
Detection of serum levels of IgE
We tested for serum IgE levels in 30 El Bagre-PF patients and in 30 control individuals from the El Bagre EPF endemic area with a commercial ELISA kit (AlaSTAT Total IgE kit, Diagnostic Products Corporation, Los Angeles, California, USA). We followed the kit instructions, and the positive cutoff level was 138 IU (international units) [8].
Direct immunofluorescence (DIF)
For DIF, 4 μm thick skin cryosections were partially fixed with paraformaldehyde, rinsed in phosphate-buffered saline (PBS, pH 6.8), incubated with fluorescein isothiocyanate (FITC) in a solubilization buffer (PBS with 0.5% Triton X-100 Octylphenolpoly (ethyleneglycolether) and then rinsed again. After blocking with PBS with 0.01%-Tween 20 (Polysorbate 20) and 0.5% bovine serum albumin (BSA), the sections were incubated with antiserum for one hour. Goat FITC conjugated anti-human IgE (Epsilon-chain) antiserum was obtained from Vector Laboratories (Burlingame, California, USA). The slides were then counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (Pierce, Rockford, Illinois, USA), washed, coverslipped and dried overnight at 4°C.
Statistical methods
Differential serum IgE levels between EPF and control samples were analyzed by the Mann-Whitney test (GraphPad Software, San Diego, California, USA). We considered p values ≤0.05 to be statistically significant.
Results
IHC results
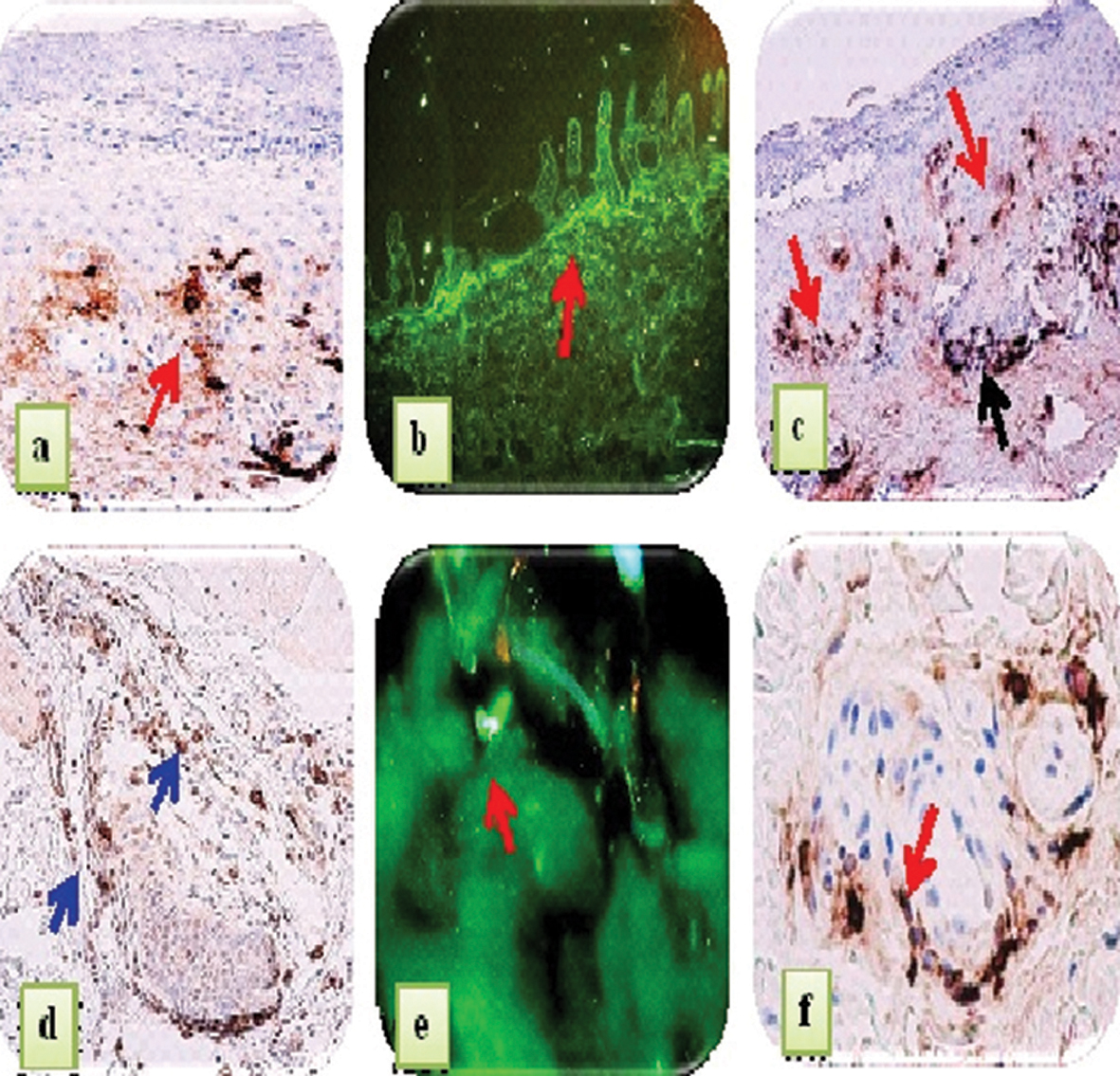
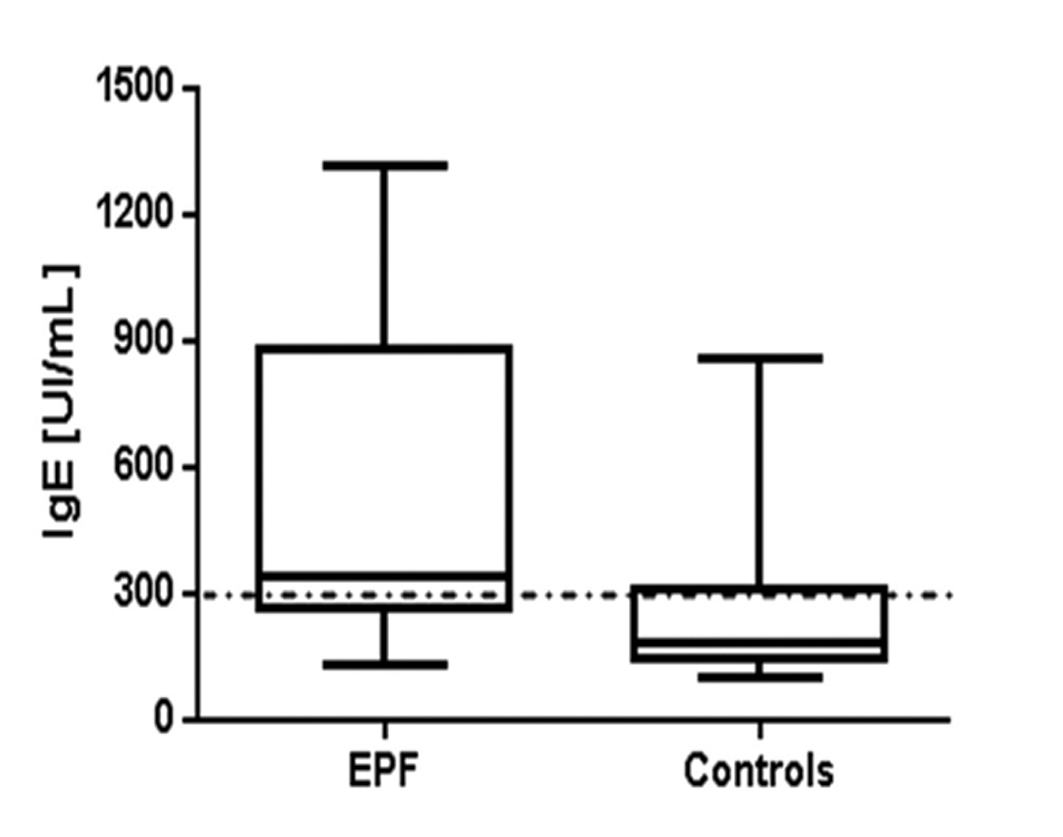
In our study, we found that MCT and c-kit were Overexpressed, primarily in perivascular infiltrates surrounding the upper dermal neurovascular plexus, as well as around dermal blood vessels supplying sebaceous and eccrine glands. As expected, normal skin demonstrated rare mast cells around these blood vessels. Interestingly, positive IgE staining was noted along the BMZ in some EPF biopsies. In eccrine gland cells, deposits of IgE were predominantly cytoplasmic, and were noted in 70% of the EPF cases. Four controls from the endemic area also stained positive for IgE, in a similar pattern as the EPF cases. Significantly, the EPF cases demonstrated stronger MCT and IgE expression than controls from the endemic and non-endemic areas. In our EPF biopsies, we demonstrated positive epidermal staining in multiple patterns utilizing the anti-IgE antibody. Some staining was intracytoplasmic, and some nuclear; most of the cases (75%) exhibited a combination of these patterns. IgE displayed positive staining within sebaceous gland sebocytes, within the base and isthmus of hair follicular units and within hair follicles. In addition to these patterns, positive staining was also noted between epidermal keratinocytes in approximately sixty (60) per cent of the EPF cases. The controls did not demonstrate these epidermal staining patterns. We also observed strong staining positivity with anti-IgE in several cases on epidermal supracorneal and subcorneal debris. Further, our MCT, c-kit and IgE staining displayed similar distributions; in several cases, the staining was seen subjacent to the basement membrane zone (BMZ) of the epidermis, as well as under the BMZs of dermal eccrine and sebaceous glands.The MCT, c-kit and IgE deposits were also seen surrounding dermal eccrine glands, blood vessels and nerves. In Figure 1, we show our IHC staining data for MCT, c-kit and IgE expression, and DIF with FITC conjugated anti-IgE in representative skin biopsies from EPF patients. In Figure 2, we shouw our IgE levels detected by ELISA testing, with increased levels in the EPF group relative to controls (p<0.0001). Table I summarizes our primary results using IHC, and the patterns of reactivity and intensity of staining of the MCT, c-kit and IgE expression in EPF and in controls. Again, the MCT, c-kit and IgE expression in PF patients displayed similar patterns and intensity.
|
Figure 1. IHC staining with anti-human MCT, IgE and c-kit, and DIF staining with FITC conjugated anti-human IgE in representative skin biopsies from EPF patients. a. IHC staining showing strong expression of MCT around upper dermal vessels (brown staining; red arrow). b. DIF showing focal linear staining at the dermal-epidermal junction with FITC conjugated IgE (green staining; red arrow). c. IHC staining with anti-IgE displays brown staining in the papillary dermis (brown staining; red arrows), as well as around the upper dermal blood vessels (brown staining; black arrow). d. IHC staining with anti-IgE shows positive staining around a hair follicle unit (brown staining; blue arrows). e. DIF with FITC conjugated IgE displaying a common pattern seen in most of the EPF cases; specifically, the presence of individual large cells (green staining; red arrow) in the dermis. f. IHC staining with c-kit/CD117 positive staining around eccrine ducts (brown staining; red arrow).
|
|
Figure 2. IgE levels detected by ELISA in EPF and control groups. The boxes represent 25 to 75 quartile ranges of values, and the horizontal lines inside the boxes the respective medians (p<0.0001).
|
Skin biopsies
|
MCT
|
c-kit/CD117
|
IgE
|
Finding conclusions
|
Brazilian EPF (n=14)
|
100% (4+) staining around
both the dermal neurovascular
plexus, and blood vessels
feeding dermal sebaceous
and eccrine sweat glands
|
80% positive staining
|
80% (4+) staining around
both the dermal neurovascular
plexus, and blood vessels
feeding dermal sebaceous and
eccrine glands
|
All samples showed high
expression compared with the
physiological baseline
|
Colombian/El Bagre EPF
(n=30)
|
100% positive (4+) staining
around the dermal neurovascular
plexus, and blood
vessels feeding dermal sebaceous
and eccrine glands
|
80% positive (3+) staining
around the dermal neurovascular
plexus, and blood
vessels feeding dermal
sebaceous and eccrine
glands
|
80% positive (4+) around the
dermal neurovascular plexus,
and blood vessels feeding
dermal sebaceous and eccrine
glands
|
All samples showed high
expression compared with the
physiological baseline
|
Controls from the endemic
area of Colombian/El
Bagre EPF (n=30)
|
4/30 (3+), 7/30 (2+), and all
others baseline physiological
staining (1+)
|
4/30 (3+), 6/30 (2+) and all
others baseline physiological
staining (1+)
|
2/30 (2+), and all others
baseline physiological staining
(1+)
|
Increased positivity relative to
plastic surgery controls and/or
Brazilian control samples
|
Normal controls from
Brazil (n=3)
|
Base line physiological staining
in all samples (1+)
|
None
|
None
|
All samples showed baseline
physiological staining compared
to the patients
|
Normal controls from
plastic surgery reductions
in Colombia and the USA
(n=15)
|
Baseline physiological staining
in all samples (1+)
|
None
|
None
|
All samples showed base line
physiological staining compared
to the patients
|
Table I. Mast cell tryptase, c-kit/CD117 and IgE immunohistochemistry(IHC) staining results in skin biopsies from endemic
pemphigus foliaceus (EPF) patients and controls.
Discussion
IgE plays a central role in Type I hypersensitivity. In response to an allergen/antigen, binding of IgE to Fcε receptors occurs on mast cells and basophils, which in turn induces signaling and leads to mast cell degranulation, immune mediator release and expression of c-kit (CD117) [14]. Mast cells reside in most tissues, including the skin, and are occasionally found around dermal blood vessels. We found stronger MCT expression in patients with EPF that in our control group. Previous authors have also demonstrated increased MCT staining in skin biopsies of patients with bullous pemphigoid [15-17]. Mast cells and basophils have several intramembranous receptors for the Fc portion of IgE (FC€RI). Aggregation of two or more of those FC€RI receptors with antigen induces cross-linking between IgE molecules. The resultant IgE-receptor activation induces complex signal transduction, leading to the release of mediators from mast cells and basophils. Pre-formed mediators from mast cell granules are released immediately, and include histamine, neutral proteases and a small number of cytokines and proteoglycans. Mast cell responsiveness can be also increased by complement; in EPF, complement plays an important role [17]. Based on our findings, we speculate that MCT, c-kit and IgE may contribute to dermal blood vessel vasodilatation, with increased permeability and leukocyte extravasation. We also speculate that in EPF, the aggregation of two or more of those FC€RI receptors with cross-linking of receptor-bound IgE molecules leads to receptor activation and complex signal transduction, thus leading to the release of mediators from mast cells and basophils. Serum IgE levels were higher in our EPF patients in comparison with controls, and this also has been shown in other autoimmune blistering diseases [17-20]. More recently, in 2013 Kalantari-Dehaghi et al. reported interesting experiments which confirm mitochondrial damage by autoantibodies in pemphigus vulgaris (PV) [21]. Moreover, in 2011 Zhang et al. investigated the role of mitochondrial dynamics in degranulation of human cultured mast cells during activation by IgE/antigen and substance P, showing that degranulation is accompanied by mitochondrial translocation from a perinuclear location to exocytosis sites. Mitochondrial translocation was also evident in skin mast cells from atopic dermatitis patients [22]. Since both forms of pemphigus (PF and PV) are endemic in Brazil [23], we speculate that common pathogenetic mechanisms may exist for both disorders. Thus, this important evidence [21,22] leads us to speculate regarding mast cells and IgE in EPF, and a mitochondrial role in the pathogenesis of this disorder. Further, it is important to note that in pemphigus and bullous pemphigoid IgG4 autoantibodies predominate, and that IgE versus IgG4 production can be differentially regulated by IL-10. Finally, timing studies (featuring acute versus chronic cases) in EPF may help to address the increased antigenic response evidenced by 1) stronger local immune expression as well as 2) the stronger seric response relative to controls.
REFERENCES
1. Diaz L, Sampaio S, Rivitti E, Martins CR, Cunha PR, Lombardi C, et al: Endemic pemphigus foliaceus (fogo selvagem). Clinical features and immunopathology. J Am Acad Dermatol. 1989;20:657-9.
2. Chiossi MP, Costa RS, Roselino AM: Dermal dendritic cell number correlates with serum autoantibody titers in Brazilian pemphigus foliaceus patients. Braz J Med Biol Res. 2004;37:337-41.
3. Chiossi MP, Roselino AM: Endemic Pemphigus foliaceus („Fogo selvagem”): a series from the Northeastern region of the State of São Paulo, Brazil, 1973-1998. Rev Inst Med Trop Sao Paulo. 2001;43:59-62.
4. Abréu-Vélez, AM, Beutner EH, Montoya F, Bollag WB, Hashimoto T: Analyses of autoantigens in a new form of endemic pemphigus foliaceus in Colombia. J Am Acad Dermatol. 2003;49:609-14.
5. Abreu-Velez, A, Hashimoto T, Bollag W, Tobón Arroyave S, Abreu-Velez CE, Londoño ML, et al: A unique form of endemic pemphigus in Northern Colombia. J Am Acad Dermatol. 2003;49:599-08.
6. Ramos W, Chacon GR, Galarza C, Gutierrez EL, Smith ME, Ortega-Loayza AG: Endemic pemphigus in the Peruvian Amazon: epidemiology and risk factors for the development of complications during treatment. An Bras Dermatol. 2012;87:838-45.
7. Morini J, Jomaa B, Gorgi Y, Saguem MH, Nouira R, Roujeau JC, et al. Pemphigus foliaceus in young women. An endemic focus in the Sousse area of Tunisia. Arch Dermatol. 1993;129:69-73.
8. Abréu-Vélez AM, Warfvinge G, Herrera WL, Abréu-Vélez CE, Montoya MF, Hardy DM, et al: Detection of mercury and other undetermined materials in skin biopsies of endemic pemphigus foliaceus. Am J Dermatopathol. 2003;25:384-91.
9. Figueiredo AM, Antunes L, Patrus OA: Hipersensibilidade cutânea mediada por IgE em pacientes com pênfigo endêmico (Fogo selvagem). (Immediate skin hypersensitivity mediated by IgE in patients with fogo selvagem). An Bras Dermatol. 1988;6:47–9.
10. Fairley JA, Burnett CT, Fu CL, Larson DL, Fleming MG, Giudice GJ: A pathogenic role for IgE in autoimmunity: bullous pemphigoid IgE reproduces the early phase of lesion development in human skin grafted to nu/nu mice. J. Invest. Dermatol. 2007;127:2605–11.
11. Dresow SK, Sitaru C, Recke A, Oostingh GJ, Zillikens D, Gibbs BF: IgE autoantibodies against the intracellular domain of BP180. Br J Dermatol. 2009;160:429-32.
12. Howard MS, Yepes MM, Maldonado JG, Villa E, Jaramillo A, Botero J, et al: Broad histopathologic patterns of non-glabrous skin and glabrous skin from patients with a new variant of endemic pemphigus foliaceus (part 1). J. Cutan Pathol. 2010;37:222-30.
13. Abreu-Vélez AM, Javier Patiño P, Montoya F, Bollag WB: The tryptic cleavage product of the mature form of the bovine desmoglein 1 ectodomain is one of the antigen moieties immunoprecipitated by all sera from symptomatic patients affected by a new variant of endemic pemphigus. Eur J Dermatol. 2003;13:359-66.
14. Eady RA, Cowen T, Marshall TF, Plummer V, Greaves MW: Mast cell population density, blood vessel density and histamine content in normal human skin. Br J Dermatol. 1979;100:623.
15. Wintroub BU, Mihm MC Jr, Goetzl EJ, Soter NA, Austen KF: Morphologic and functional evidence for release of mast-cell products in bullous pemphigoid. N Engl J Med. 1978; 23;298:417-21.
16. Borrego L, Maynard B, Peterson EA, George T, Iglesias L, Peters MS, et al: Deposition of eosinophil granule proteins precedes blister formation in bullous pemphigoid comparison with neutrophil and mast cell granule proteins. Am J Pathol. 1996;148:897-909.
17. Klapholz L, Kupietzky A, Weinrauch L, Shalit M, Okon E: Increased numbers of mast cells in pemphigus vulgaris skin lesions. A histochemical study. Acta Derm Venereol. 1991;71:269–71.
18. Bruns GR, Ablin RJ, Guinan PD: Serum immunoglobulin E in pemphigus. J Invest Dermatol. 1978;71:217–8.
19. Nagel A, Lang A, Engel D, Podstawa E, Hunzelmann N, de Pita O et al: Clinical activity of pemphigus vulgaris relates to IgE autoantibodies against desmoglein 3. Clin Immunol. 2010;134:320-30.
20. Qian Y, Prisayanh P, Andraca E, Qaqish BF, Aoki V, Hans-Filhio G, et al: IgE, IgM, and IgG4 anti-desmoglein 1 autoantibody profile in endemic pemphigus foliaceus (fogo selvagem). J Invest Dermatol. 2011;131:985-7.
21. Kalantari-Dehaghi M, Chen Y, Deng W, Chernyavsky A, Marchenko S, Wang PH, et al: Mechanisms of mitochondrial damage in keratinocytes by pemphigus vulgaris. J Biol Chem. 2013;288:16916-25.
22. Zhang B, Alysandratos K-D, Angelidou A, Asadi S, Sismanopoulos N, Delivanis D-A, et al: Human mast cell degranulation and preformed TNF secretion require mitochondrial translocation to exocytosis sites: Relevance to atopic dermatitis. Allergy Clin Immunol. 2011;127:1522–31.
23. Gonçalves GA, Brito MM, Salathiel AM, Ferraz TS, Alves D, Roselino AM: Incidence of pemphigus vulgaris exceeds that of pemphigus foliaceus in a region where pemphigus foliaceus is endemic: analysis of a 21-year historical series. An Bras Dermatol. 2011;86:1109-12.
Comments are closed.