Ganglionic melanoma without cutaneous localization: primitive, or metastasis?
Asmae Rasso , Zakia Douhi, Sara Elloudi, Hanane Baybay, Fatima Zahra Mernissi
, Zakia Douhi, Sara Elloudi, Hanane Baybay, Fatima Zahra Mernissi
Department of Dermatology CHU Hassan II, Fez, Morocco
Corresponding author: Dr. Asmae Rasso
Submission: 20.03.2020; Acceptance: 04.05.2020
DOI: 10.7241/ourd.2020e.175
Cite this article: Rasso A, Douhi Z, Elloudi S, Baybay H, Mernissi FZ. Ganglionic melanoma without cutaneous localization: primitive, or metastasis?. Our Dermatol Online. 2020;11(e):e175.1-e175.3.
Citation tools:
Copyright information
© Our Dermatology Online 2020. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
The ganglionic melanoma without cutaneous primitive is a rare entity, and that constitutes a challenge for the dermatologist, which pushes him to repeat his dermatological examination several times while searching for the primitive. Case report: We describe the case of a patient aged 73 years, with asymptomatic left inguinal swelling. Dermatologic examination found vitiligo-like leukoderma with a 2% body surface, and 2 cm left inguinal lymphadenopathy, with no inflammatory signs. Histology and immunohistochemistry favored ganglionic melanoma. The complete assessment did not reveal other metastases. the patient benefited from extensive excision with radiotherapy and then chemotherapy without recurrence. Conclusion: ganglionic melanoma without mucocutaneous primitive suggests a high probability of cutaneous regression of a metastatic melanoma, especially if the patient presents a vitiliginous reaction.
Key words: Ganglionic; Melanoma; Vitiligo-like leukoderma; PET scanner; mutation anti-BRAF
INTRODUCTION
Melanoma with unknown primary site, described for the first time in 1952 [1], is defined as histologically confirmed subcutaneous, lymph nodal or visceral metastatic melanoma with no evidence of primary lesion.Melanoma is developed from melanocytes, Although most develop on sunlight-exposed areas, this disease can also developon other areas. Melanomas of the eye, ear, vaginale epthelium, meninges are well documented but rare. They account for less than 10% of all melanomas [2]. we report a patient with lymphadenopathy melanoma with no known primary site.
CASE REPORT
A 73-year-old patient, without any notable pathological history, consulted after discovery of an asymptomatic inguinal mass, appeared over 1 month, without any other signs associated. The examination of the ganglion air found left inguinal lymphadenopathy, 3 cm in size, without inflammatory signs. Dermatologic examination revealed achromic macules on the back, the legs, and genital, with a body surface area of ??3% (Fig. 1), and with contrast enhancement in “Wood’s light”. And revealed three dermal nevus not suspect clinically, seat at the neck and back. The rest of somatic examination was normal, with a general state preserved. Ultrasonography of the ganglionic areas found hypoechogenic left inguinal adenopathies of 25mm for the largest central necrosis and anarchic vascularization. The biopsy of adenopathy was made, of which histology and immunohistochemistry (HMB 45+, S100 +, Melan A +), was in favor of ganglionic melanoma. The mutaton anti-BRAF and C-KIT was negative.
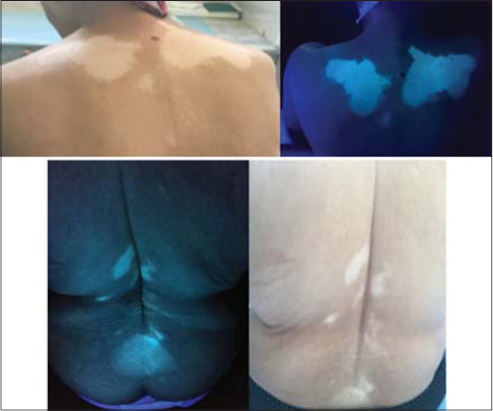 |
Figure 1:Vitiligo-like leukoderma with body surface area 3%. |
The search for the primitive by the ophthalmologic examination, cervico-vaginal smear, ano-recto-colonoscopy, as well as a cerebral CT did not bring any element in favor of melanoma. The cervical-thoracic-abdominopelvic CT showed no other metastatic locations. The biological assessment was normal. Then a PET scanner showed a subcutaneous nodule opposite the anterior surface of the tibial plateau weakly hyper-metabolic (SUVmax0,7) An excision of this lesion did not objectify a cutaneous melanoma.
The patient was treated by inguinal lymphadenopathy surgery, and radiotherapy (30gr fraction over 5 sessions) (Fig. 2). Four months after treatment, the cervical-thoracic-abdominopelvic CT showed the appearance of others lymphadenopathy internal iliac with central necrosis. then the patient was treated by chemotherapy with size stabilization of the lymphadenopathy on the control scanner, after 6 months of follow-up, without relapse.
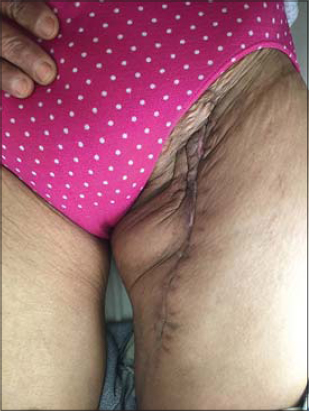 |
Figure 2:Linear inguinal scar. |
DISCUSSION
Melanoma of lymph nodes with unknown primary site is a rare phenomenon with an incidence of 3.2% [3]. The extra-cutaneous melanoma can come from the uvea, leptomening, mucous membranes and from anywhere in the digestive tract. including gall bladder. It occurs at any age but especially the patients of fourth and fifth decades. It is twice as common in humans. The diagnosis of certainty is histological, confirmed by the Fontana-Masson staining, and the immunohistochemistry (S100, HMB 45). The ganglionic melanoma suggests more a metastatic cutaneous-mucosal primitive, with a complete regression, than a lymph node primitive, seen absence of melanocytes at the ganglion level, and especially if the patient has a vitiligo-like leukoderma associate, as the case of our patient [4]. Melanoma regression is rare, with an incidence of 0.22% to 0.27%. it can be partial in 46%, or complete in 10% of cases, sometimes rapidly. This regression potentially occurs via a tumor specific natural cytolytic T cell response, which explains the vitiliginous response [5]. Although the cause of vitiligo-like leukoderma is still unclear, in most situations, but all know that it is an autoimmune disease in which melanocytes are destroyed by antigen-specific T cells. The association vitiligo-like leukoderma and melanoma is described in the literature with an incidence of 2.8%, this association is explained by the fact that melanoma shares several antigens with normal melanocytes including Melan-A/MART-1, tyrosinase and gp100, which leads to a cross-immune reaction [6,7].
The extension assessment is systematic in the melanoma but when normal with a nodal ganglionnaire a systematic pet scanner can be discussed because it this paraclinical “modern” gives a good sensitivity and specificity [8]. The pet scanner of our patient revealed a pre-tibial hypersensitivity whose histology was not specific, but it does not eliminate the regression of melanoma, especially since the lesion sits on the same limb of the lymadenopathy.
CONCLUSION
our case cited here support the hypothesis that lymph node melanoma without obvious primitive suggests a high probability of metastatic melanoma with complete regression, especially if the patient has a vitiliginous immune response.
Consent
The examination of the patient was conducted according to the Declaration of Helsinki principles.
The authors certify that they have obtained all appropriate patient consent forms. In the form the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
REFERENCES
1. Forbes W, Maloney AFJ. Primary melanomatosis of the leptomeninx. Pathal Bacterial. 1950;62;403-9.
2. Bebe FN, Hu S, Brown TL, Tulp OL. Metastatic melanoma in Florida, 1996-2010:Racial, demographic, occupational and tumor characteristics, and burden of metastasis. Our Dermatol Online. 2018;9:369-79.
3. Wohlmuth C, Wohlmuth-Wieser I, May T, Vicus D, Gien LT, Laframboise S. Malignant melanoma of the vulva and vagina:A US population-based sstudy of 1863 Patients. Am J Clin Dermatol. 2020;21:285-95.
4. Bae JM, Choi YY, Kim DS, Lee JH, Jang HS, Lee JH, et al. Metastatic melanomas of unknown primary show better prognosis than those of known primary:a sys- tematic review and meta-analysis of observa- tional studies. J Am Acad Dermatol. 2015;72:59–70.
5. Saàdani CH, Gallouj S, Zinoune S, Senhaji G, Baybay H, Mernissi FZ, et al. Diffuse melanosis cutis secondary to metastatic malignant melanoma:case report. Our Dermatol Online. 2019;10:79-81.
6. Francisco G, Rao BK, Victor FC. Two reports of malignant melanoma arising within a new vitiligo-like depigmented patch. JAAD Case Rep. 2018;4:573-5.
7. Aslam I, Konopinski J, Aziz N. Metastatic melanoma masquerading as a furuncle. Our Dermatol Online. 2018;9:434-7.
8. Shivamurthy A, Kudva R, Kudva A, Jaiprakash P. Metastatic spindle cell melanoma on cytology –a diagnostic challenge. Our Dermatol Online. 2019;10:159-62.
Notes
Source of Support: Nil.
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/000-0003-3455-3810 http://orcid.org/000-0003-3455-3810 |



Comments are closed.