How narrow-band and broad-band UVB irradiation influences the immunohistochemistry analyses of experimental animals’ skin – a comparative study. Part II.
Katarzyna Borowska
Department of Histology and Embryology with Experimental Cytology Unit, Medical University of Lublin, 11 Radziwiłłowska, 20–080 Lublin, Poland
Corresponding author: Prof. Katarzyna Borowska, E-mail: k_borowska@wp.pl
Submission: 14.09.2017; Acceptance: 28.09.2017
How to cite this article: Borowska K. How narrow-band and broad-band UVB irradiation influences the immunohistochemistry analyses of experimental animals’ skin – a comparative study. Part II. Our Dermatol Online. 2017;8(3e):e6.
ABSTRACT
This is the second part of the artcle series impact narrow-band UVB radiation (NB-UVB) and broad-band UVB radiation (BB-UVB) on experimental animals’ skin (white Wistar female rats). The aim of this comparative study was immunohistochemistry analyses containing expression of p53 protein. Expression of p53 protein was performed on two experimental groups. One – exposed to NB-UVB; the other – exposed to BB-UVB radiation. The results indicate that p53 protein takes an active part in the process of apoptosis that is induced by both NB-UVB and BB-UVB. The results showed an increase in p53 expressing cells following BB-UVB than NB-UVB phototherapy.
Key words: narrow-band UVB, TL01, 311 nm, broad-band UVB, TL 12, p53 protein.
INTRODUCTION
This is the second part of the artcle series impact narrow-band UVB radiation (NB-UVB) (311 nm) and broad-band UVB radiation (BB-UVB) (290-320 nm) on experimental animals’ skin. The aim of this comparative study was immunohistochemistry analyses containing expression of p53 protein.
MATERIAL AND METHODS
Expression of p53 protein was performed on two experimental groups. One – exposed to NB-UVB; (D- dorsal part and A – abdominal part) the other – exposed to BB-UVB radiation (D- dorsal part and A – abdominal part). The experimental animals were white Wistar female rats. The control group and experimental groups were described in detail in my previous articles – part I [1].
Immunohistochemical analyses for p53 protein was done using standard protocols. IHC tests were then done using antibodies directed against protein 53 (P5813; Sigma-Aldrich, St. Louis, MO, USA; dilution 1:100). The exposure of the antigenic sites was performed thermally by incubation in citrate buffer solution with pH = 6, in a microwave oven at 800 W, for 3 cycles lasting 5 min. The material was incubated in a primary antibody diluted as recommended by the manufacturer overnight at 4 °C. To visualize the reaction, diaminobenzidine solution (DAB) and hematoxylin colouration were used. In the negative control, experiments were conducted in a similar manner, but omitted the specific primary antibody. The material was evaluated with the use of a light microscope using objective×20 and×40. The percentage of cells with positive expression was calculated by counting approximated 300 cells in at least three randomly chosen microscopic fields (objective 40). The intensity of expression of individual proteins of cutaneous cells was compared. The intensity was graded as low (+), intermediate (++) and high (+++), by using the BX4 image analysis system manufactured by Olympus, with a DP 25 digital camera and the Cell^D software. The percentage of cells of the given intensity expression in each analyzed fields was calculated. These values were expressed as a mean ± standard deviation. The obtained tests results were subjected to statistical analysis with the use of Statistica 10.0 software StatSoft Polska. The chi-square (χ2) test was applied to compare the mean of proteins indicating positive and negative cells in groups.
Control group
Expression of p53 protein revealed normal epidermis and dermis.
Experimental group – NB-UVB-D
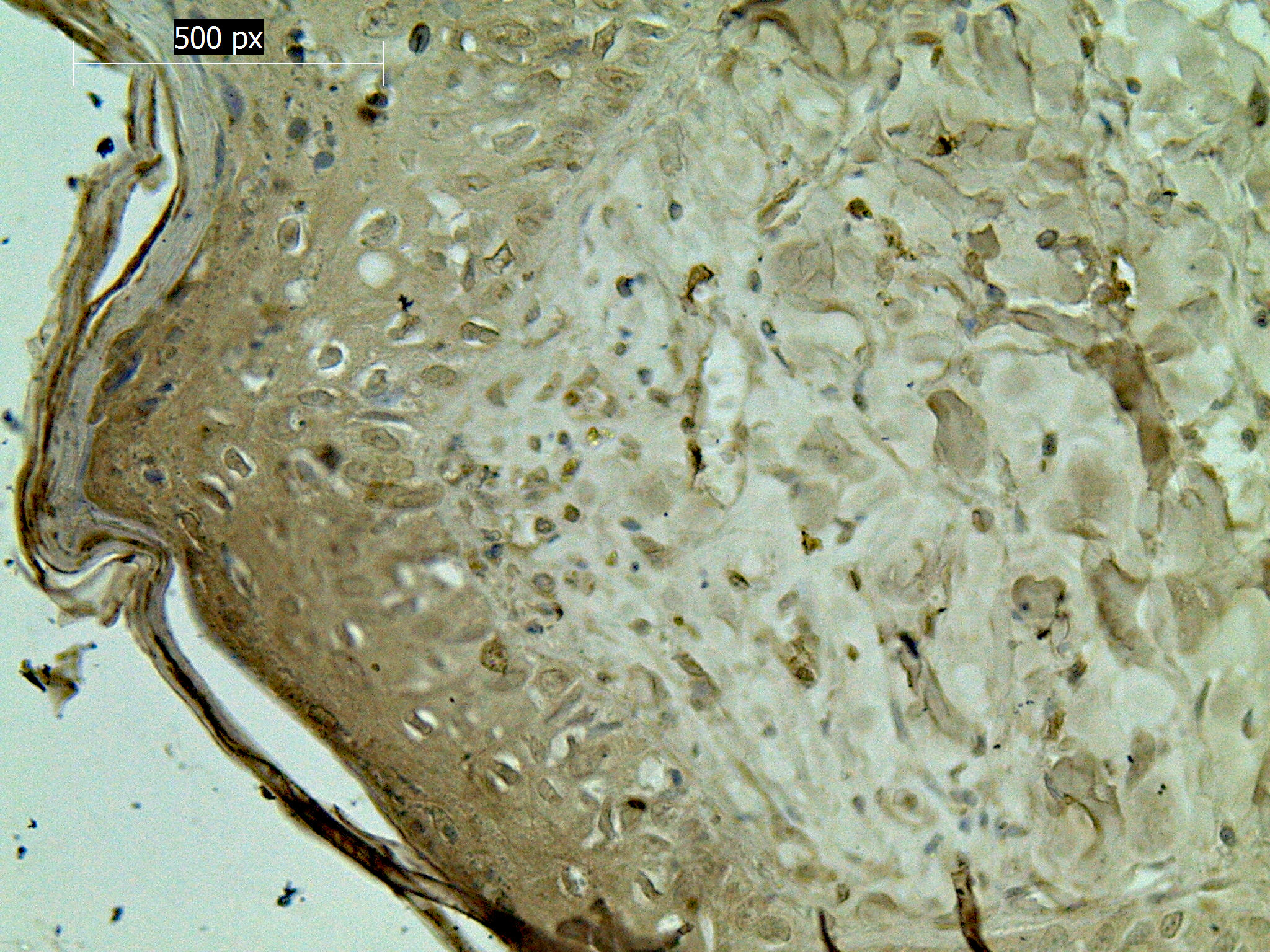
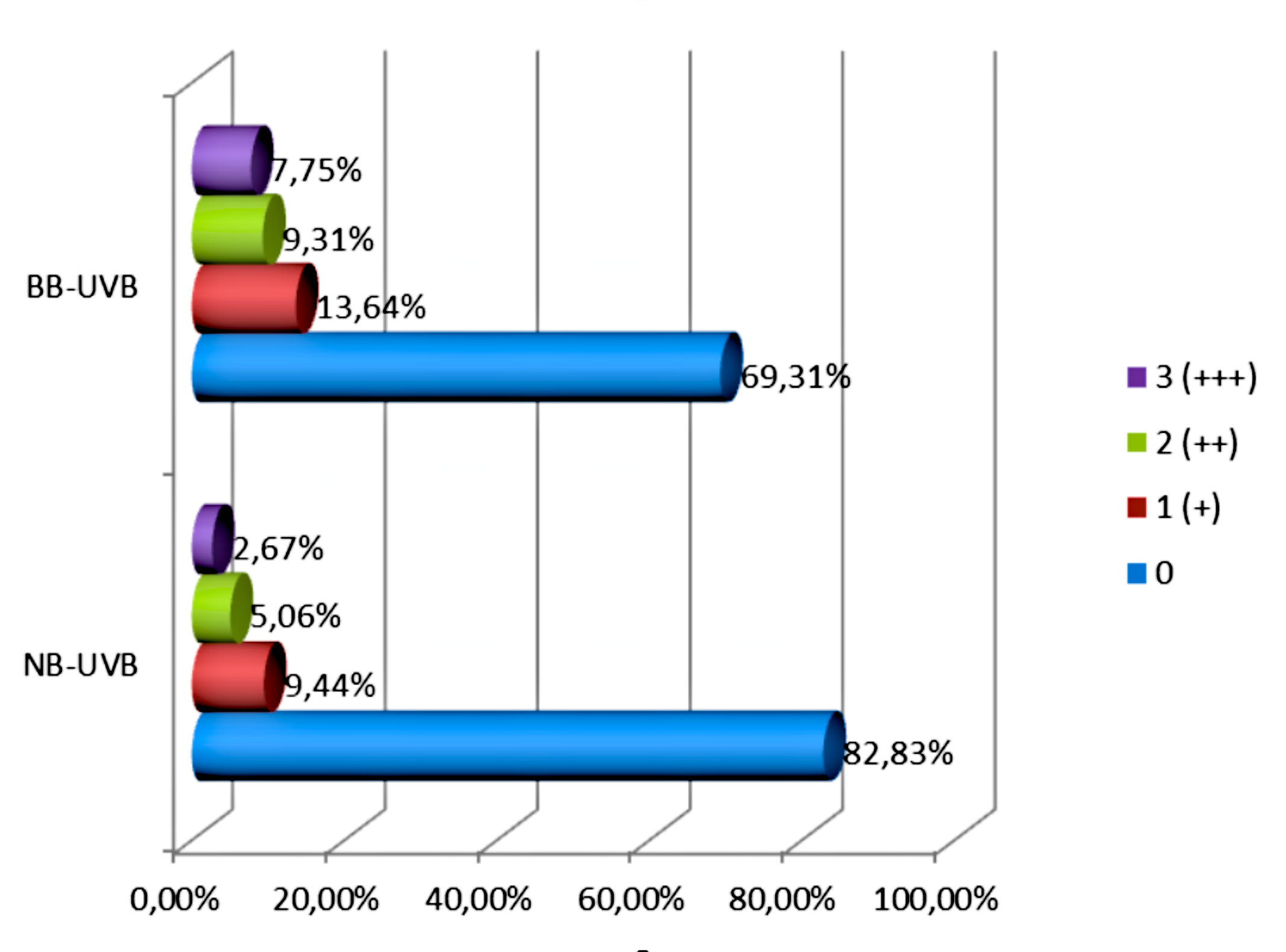
The intensity of the p53 protein expression of NB-UVB group was quite low. The high expression (a rating of 3 [+++]) was observed in 9.44%. The average expression (a rating of 2 [++]) was observed in 5.06%, while the expression evaluated as 1 [+] appeared in only 2.67%. In the cells of skin rats treated with NB-UVB, the expression of p53 protein was observed in the cell nuclei (Fig. 1).
Experimental group – NB-UVB-A
Expression of p53 protein revealed the epidermis and dermis were normal. Their appearance was similar to that in the control group animals.
Experimental group – BB-UVB-D
The high expression of p53 protein (a rating of 3 [+++]) was observed in 13.64%. The average expression (a rating of 2 [++]) was observed in 9.31%, while the expression evaluated as 1 [+] appeared in only 7.75%. In the cells of skin rats treated with BB-UVB, the expression of p53 protein was observed in the cell nuclei (Fig. 2).
Experimental group – BB-UVB-A
Expression of p53 protein revealed the epidermis and dermis were normal. Their appearance was similar to that in the control group animals.
Statistically results
There was a statistically significant relationship reaction (expression of p53 protein) and study group (p <0.0001). Statistically significant relationship between the intensity of immunohistochemical reaction and study group was also observed. (p <0.0001). No intensity of the immunohistochemical reaction in the control group occurs in intensity about 99% and in the NB-UVB group is approximately 69%. There was a statistically significant relationship between the intensity of immunohistochemical reaction and study group (p <0.0001). The expression of p53 protein of defined intensity in particular research groups presents figure 3. The value of the Pearson function: χ^2=205.323; p<0.0001.
DISCUSSION
NB-UVB and BB-UVB at equivalent MED doses were equally potent in inducing p53 expression as well as apoptosis in healthy volunteers [2]. p53 protein plays an important role in skin cancer prevention through its regulation of apoptosis in response to UVB radiation-induced DNA damage. In response to DNA damage, p53 protein accumulates in cells, translocates to the nucleus, activates gene transcription, and ultimately results in cell cycle arrest, which is thought to permit repair of damaged DNA prior to cell replication [3-5]. A positive correlation between histomorphological changes and immunohistochemistry analyses (expression of p53 protein) of experimental animals’ skin was found in the my investigation [1]. The results indicate that p53 protein takes an active part in the process of apoptosis that is induced by both NB-UVB and BB-UVB. The results showed an increase in p53 expressing cells following BB-UVB than NB-UVB radiation. The high expression of p53 protein (a rating of 3 [+++]) in BB-UVB group was observed in 13.64%. The high expression in NB-UVB group was observed in 9.44%. The expression of p53 protein in both groups was observed in the cell nuclei. Jasim ZF et al [6] also showed an increase in p53 expressing keratinocytes following NB-UVB phototherapy. Some of cells were arranged spatially, as conical clones arising from putative stem cell compartments, suggesting that the chronic NB-UVB phototherapy might have triggered cell growth and clonal expansion. In conclusions, the results showed an increase in p53 expressing cells following BB-UVB than NB-UVB phototherapy.
REFERENCES
1. Borowska K. How Narrow-Band and Broad-Band UVB Irradiation Influences the Histomorphology Evaluations of Experimental Animals’ Skin – a Comparative Study. Part I. Our Dermatol. Online. 2017;8:e5.
2. Tjioe M, Smiths T, van de Kerkhof PCM, Geritsen MJP. The differential effect of broad band vs. narrow-band UVB with respect to photodamage and cutaneous inflammation. Exp. Dermatol. 2003;12:729–33.
3. Tron VA, Trotter MJ, Tang L, Krajewska M, Reed JC, Ho VC, et al. p53-regulated apoptosis is differentiation dependent in ultraviolet B-irradiated mouse keratinocytes. Am J Pathol. 1998;153:579–85.
4. Kramata P, Lu YP, Lou YR, Singh RN, Kwon SM, Conney AH. Patches of mutant p53-immunoreactive epidermal cells induced by chronic UVB Irradiation harbor the same p53 mutations as squamous cell carcinomas in the skin of hairless SKH-1 mice. Cancer Res. 2005;65:3577–585.
5. Petitjean A, Mathe E, Kato S, Ishioka C, Tavtigian SV, Hainaut P, et al. Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat. 2007;28:622–9.
6. Jasim ZF, Lioe TF, McKenna KE, Robson T, Ouhtit A. The effect of ultra violet B (TL-01) phototherapy on epidermal expression of p53 protein in psoriatic plaques. Photodermatol Photoimmunol Photomed. 2006;22:12-7.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.

Comments are closed.