Light-emitting diode therapy for the management of radiodermatitis
Fouzia Hali1, Asmaa El Kissouni 1, Soumiya Chiheb1, Meryem Charkaoui2, Yasmina Berreda2, Nadia Benchakroune2, Souha Sahraoui2
1, Soumiya Chiheb1, Meryem Charkaoui2, Yasmina Berreda2, Nadia Benchakroune2, Souha Sahraoui2
1Department of Dermatology and Venereology, HUC Ibn Rochd, University Hassan II of Casablanca, Morocco, 2Mohamed VI Cancer Treatment Center, HUC Ibn Rochd, University Hassan II of Casablanca, Morocco
Citation tools:
Copyright information
© Our Dermatology Online 2023. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Background: Light-emitting diode (LED) is a process by which specific sequences of low-energy light are employed to regulate cellular activity without a thermal effect. The aim of this study was to evaluate the use of LED light in the treatment of radiodermatitis.
Materials and Methods: This was a prospective study that included twenty patients with chronic or acute radiation dermatitis regardless of the grade or underlying neoplasia. All these patients received two LED sessions per week according to the following protocol: 660 + 850 nm, total fluence: 36 J/cm2, total energy: 21.6 KJ. The evolution was judged from the data of the clinical examination and photography.
Results: Three patients had chronic radiodermatitis (CRD) and seventeen patients had acute radiodermatitis (ARD). The lesions appeared after ten sessions on average: a cumulative dose of 26.7 Gy. Evolution after LED treatment: For CRD, no significant improvement was noted. For ARD, regression of erythema and epidermalization of the lesions were observed after an average of six sessions (2–8 sessions).
Discussion: Vitro studies have demonstrated that LED light accelerates wound healing by increasing procollagen synthesis and decreasing inflammatory mediators. In our study. we noticed an improvement in patients with ARD regardless of the site or the underlying neoplasia. However, no improvement was observed in patients with CRD.
Conclusion: LED therapy may be an effective therapeutic option in the management of ARD, yet studies with a larger sample are necessary.
Key words: LED Light; Photobiomodulation; Radiodermatitis; Acute Radiodermatitis; Radiotherapy
INTRODUCTION
After Marie Curie’s discovery of radium, the invention of radiotherapy marked the beginning of a new era in medicine, giving a new glimmer of hope to patients with cancer. Nevertheless, one of its most frequent and debilitating side effects is radiodermatitis (RD). In fact, 95% of patients with cancer who receive radiotherapy may experience RD in some form, including erythema, dry scaling, and moist scaling [1,2]. These skin reactions to radiation cause a myriad of problems that delay treatment and lower the quality of life.
Light emitting diodes (LEDs) are sophisticated semiconductors that transform electrical current into incoherent, narrow-spectrum light, which may be absorbed by molecular chromophores or photoreceptors on the skin and activate the mitochondrial respiratory chain, triggering a cascade of cellular reactions with no thermal effect [3,4].
The aim of this study was to assess the effectiveness of LED light in the management of radiodermatitis.
MATERIALS AND METHODS
From June 2021 to January 2022, we conducted a prospective study at our dermatology department in partnership with a cancer treatment center. All patients included had histologically proven cancer for which radiotherapy was required and who had developed RD as a side effect.
Patients with RD, regardless of chronicity or severity, were recruited for this study, except minors, pregnant and breastfeeding females, and patients with a photosensitizing condition or a personal history of melanoma or lupus.
After receiving explicit written consent from the patients and performing an initial clinical evaluation as well as the staging of the RD, we used the Triwings® device (Biophoton SRL, Saint Alban, France) twice a week according to the following protocol: 660 + 850 nm, total fluence: 36 J/cm2, total energy: 21,6 KJ, total time per session: 8 minutes.
The evolution was assessed with the data from the clinical examination and photography, such as the regression of skin lesions and functional symptoms such as pain, as well as the patient’s satisfaction.
Ethics Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
RESULTS
Study Population
This study included twenty patients, with an average age of 50.4 years, ranging from 31 to 70 years. There was a significant female predominance, with a sex ratio (M/F) of 0.2. Among these patients, fifteen were monitored for breast cancer, four for cavum cancer, and one for cervical cancer. Thus the sites irradiated were the breast and axillary area (n = 15), the cervical region (n = 4), and the pelvis (n = 1). Each of these patients received between 15 and 40 radiotherapy sessions, with an average of 30. The total dose administered ranged between 40 and 70 Gy, with an average of 2.67 Gy per session.
In our study group, three patients had chronic radiodermatitis (CRD), whereas seventeen had acute radiodermatitis (ARD), among which nine had grade 1 ARD, five had grade 2 ARD, and one had grade 3 ARD. Radiodermatitis lesions emerged after an average of ten sessions, resulting in a cumulative dose of 26.7 Gy. It ought to be highlighted that none of the patients’ radiotherapy sessions were interrupted. In fact, three patients received LED sessions simultaneously with their radiotherapy sessions, while seventeen patients began LED sessions after the radiotherapy sessions ended.
Evolution after LED Sessions
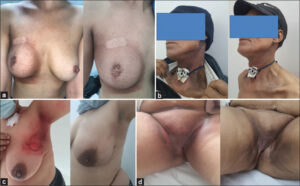
When assessing the evolution of patients after LED sessions, we noted no significant improvement in patients with CRD (Fig. 1a). While, for patients with ARD, an improvement was noted over the course of the sessions. In fact, for patients with grade 1 ARD, a regression of erythema and skin whitening was observed after an average of three sessions (2–5 sessions) (Fig. 1b). Meanwhile, in those with grade 2 ARD, the epidermalization of the lesions was observed after an average of six sessions (4–8 sessions) (Figs. 1c and 1d). On the other hand, the patient with grade 3 ARC healed her lesions after eight sessions of LED light.
DISCUSSION
Radiodermatitis is defined as any skin reaction caused by ionizing radiation (photons, electrons, and so forth) [1,5]. In terms of appearance, undifferentiated basal layer keratinocytes are radiosensitive: ionizing rays cause an inflammatory reaction and oxidative stress (OS), which interact and reinforce each other, leading to radiation-induced cellular damage and even cell death. In the chronic phase, inflammation and OS may cause alterations in numerous cytokines, cell cycle modifications, and DNA damage, sustaining the cascade and leading to late responses [1,6,7].
ARD occurs within days or weeks of radiation exposure. There are three subtypes of ARD: dry radiodermatitis, exudative radiodermatitis, and acute radionecrosis. A severity classification of five grades was proposed by the European Organization for Research and Treatment of Cancer [8] (Table 1). CRD, on the other hand, develops months or even years after irradiation and worsens over time, with no relation to the severity of ARD. There are several subtypes: radiodystrophy, late radionecrosis, and cancer, emphasizing the significance of lifelong surveillance [9–11].
LED light has been demonstrated to stimulate fibroblasts and play a role in expediting and enhancing wound healing. Indeed, in vitro studies have demonstrated that LED light accelerates wound repair, enhances procollagen production, decreases inflammatory mediators, and decreases dermal matrix metalloproteinase (MMP) in cultured fibroblasts exposed to irradiation [12], motivating various authors to test LED light against the treatment of radiodermatitis [13,14]. Thus, Camargo et al. [15] investigated the effect of LED light in an experimental model of radiodermatitis in ten rats. The animals were irradiated with 20 Gy in a single session and, fifteen days later, they were divided into two groups: a control group (five rats) and a group receiving LED sessions (660 nm) every two days for twenty-one days. The authors observed a clinical and even histological improvement in the LED group, since histologically they noticed that LED sessions improved the division and migration of cells in the basal layer of the epidermis, thus increasing the speed of epithelialization, which proved the regenerative potential of this treatment.
The efficacy of LED has been also evaluated in radiodermatitis prevention. In fact, Strouthos et al. [16] conducted a prospective study with a control group of seventy patients with breast cancer treated with radiotherapy following conservative surgery. The study revealed that LED light sessions (660 + 850 nm) applied twice a week before radiotherapy could be beneficial in minimizing the incidence and sequelae of radiation-induced skin damage as well as pain in patients with breast cancer.
LED light therapy was employed not only in patients with breast cancer yet also in patients with head and neck cancer (HNC). In one case report study, two HNC patients had grade 3 radiodermatitis following RT treatment. LED light was employed every day following a protocol (660 nm, 27.77–35.71 J/cm2, 40–100 mW). After 48 hours, one of the patients’ skin responses improved. The second patient whitened entirely after seven LED light sessions [17], which was similar to our findings.
To the best of our knowledge, our study was the first to assess the efficiency of LED light in the management of RD, regardless of the type/grade or the underlying neoplasia. We witnessed a marked improvement and healing of lesions in ARD after 2–8 sessions of LED light. Healing was faster in grade 1 ARD, while grade 3 ARD required more sessions, yet the overall evolution was favorable regardless of the site of irradiation. Nevertheless, no improvement was observed in CRD.
CONCLUSION
RD is a significant handicap that affects the patient’s quality of life and poses a major management challenge. Despite therapeutic advances and a plethora of treatment options proposed, few therapies have proven to be effective in the management of RD. In this study, we found that LED light therapy could be a beneficial option for patients with ARD, given its effectiveness, safety, ease of use, and low cost. However, additional prospective studies, particularly, randomized, double-blind studies with a larger sample size, are required to confirm these findings.
Statement of Human and Animal Rights
All the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the 2008 revision of the Declaration of Helsinki of 1975.
Statement of Informed Consent
Informed consent for participation in this study was obtained from all patients.
REFERENCES
1. Ginex PK, Backler C, Croson E, Horrell LN, Moriarty KA, Maloney C, et al. Radiodermatitis in patients with cancer:Systematic review and meta-analysis. Oncol Nurs Forum. 2020;1:225-36.
2. Bray FN, Simmons BJ, Wolfson AH, Nouri K. Acute and chronic cutaneous reactions to ionizing radiation therapy. Dermatol Ther. 2016;6:185-206.
3. Barolet D. Light-emitting diodes (LEDs) in dermatology. Semin Cutan Med Surg. 2008;27:227-38.
4. Oh PS, Jeong HJ. Therapeutic application of light emitting diode:Photo-oncomic approach. J Photochem Photobiol B. 2019;192:1-7.
5. Singh M, Alavi A, Wong R, Akita S. Radiodermatitis:A review of our current understanding. Am J Clin Dermatol. 2016;17:277-292.
6. Hymes SR, Strom EA, Fife C. Radiation dermatitis:Clinical presentation, pathophysiology, and treatment. J Am Acad Dermatol. 2006;54:28-46.
7. Wei J, Meng L, Hou X, Qu C, Wang B, Xin Y, et al. Radiation-induced skin reactions:Mechanism and treatment. Cancer Manag Res. 2019;11:167-77.
8. Russi EG, Bensadoun R-J, Merlano MC, Bourhis J, Ricardi U, Giralt J, et al. Bio-radiation dermatitis:The need of a new grading, Ann Oncol. 2013;24:2463-5.
9. Robijns J, Laubach H-J. Acute and chronic radiodermatitis:Clinical signs, pathophysiology, risk factors and management options. J Egypt Womenʼs Dermatol Soc. 2018;15:2-9.
10. Spałek M. Chronic radiation-induced dermatitis:Challenges and solutions. Clin Cosmet Investig Dermatol. 2016;9:473-82.
11. Davis MM, Hanke CW, Zollinger TW, Montebello JF, Hornback NB, Norins A. Skin cancer in patients with chronic radiation dermatitis. J Am Acad Dermatol. 1989;20:608-16.
12. Chang L-Y, Fan SM-Y, Liao Y-C, Wang W-H, Chen Y-J, Lin S-J. Proteomic analysis reveals anti-fibrotic effects of blue light photobiomodulation on fibroblasts. Lasers Surg Med. 2020;52:358-72.
13. Robijns J, Lodewijckx J, Mebis J. Photobiomodulation therapy for acute radiodermatitis. Curr Opin Oncol. 2019;31:291-8.
14. Posten W, Wrone DA, Dover JS, Arndt KA, Silapunt S, Alam M. Low-level laser therapy for wound healing:Mechanism and efficacy. Dermatol Surg. 2005;31:334-40.
15. Camargo CP, Carvalho HA, Maluf FC, Sousa A, Perin P, Perin M, et al. Light-emitting diode stimulates radiodermatitis recovery. Acta Cir Bras. 2021;20:608-16.
16. Strouthos I, Chatzikonstantinou G, Tselis N, Bon D, Karagiannis E, Zoga E, et al. Photobiomodulation therapy for the management of radiation-induced dermatitis:A single-institution experience of adjuvant radiotherapy in breast cancer patients after breast conserving surgery. Strahlenther Onkol. 2017;193:491-8.
17. Rocha BA, Simões A, Lima LMC, Teixeira MMS, Martinez AS, Melo MR, et al. Treating acute cervical radiodermatitis with photobiomodulation therapy:A report of two cases. Photobiomodul Photomed Laser Surg. 2020;38:19-23.
Notes
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
|




Comments are closed.