Severity of acne and quality of life of patients treated at Cameroonian hospitals (sub-Saharan Africa)
Emmanuel Armand Kouotou1,2, Ulrich Nguena Feungue 3, Dahlia Noelle Tounouga1,4, Audrey Sandra Ngoune Madjoukeng2, Grâce Anita Nkoro2, Berline Odette Sigha5, Rose Kotto Ekambi6, Anne Cécile Zoung-Kanyi Bissek2
3, Dahlia Noelle Tounouga1,4, Audrey Sandra Ngoune Madjoukeng2, Grâce Anita Nkoro2, Berline Odette Sigha5, Rose Kotto Ekambi6, Anne Cécile Zoung-Kanyi Bissek2
1Yaoundé Central Hospital, Cameroon, 2Faculty of Medicine and Biomedical Sciences, Yaoundé, Cameroon, 3University Teaching Hospital of Treichville, Abidjan, Ivory Coast, 4Faculty of Health Sciences, Cotonou, Benin, 5Faculty of Health Sciences, Bamenda, Cameroon, 6Faculty of Medicine and Pharmaceutical Sciences, Douala, Cameroon
Citation tools:
Copyright information
© Our Dermatology Online 2023. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Background: The aim of this study was to assess the severity of acne and the impact on the quality of life (QoL) of patients undergoing anti-acne treatment.
Methods: This was a cross-sectional study conducted from January to April 2017 at three hospitals in Yaoundé. Patients suffering from acne and under treatment were consecutively included. Clinical severity and QoL were assessed with the ECLA and CADI scales, respectively. Data was analyzed with SPSS 23.0. We employed the ANOVA test and linear regression to search for associations between variables. The significance level was at 5%.
Results: We recruited 113 patients, predominantly female (83%), with a mean age of 26.0 ± 6.4 years. Mild-to-moderate forms were predominant (74%). The mean ECLA score was 10.1 ± 4.6, with a majority of the participants 74.3% (84/113) having mild-to-moderate acne, while 25.7% (29/113) had severe acne. The majority of the patients found the treatment expensive (69%). Therapeutic education had been conducted in 46.9% of the patients, and 60.2% of them reported a relapse during treatment. An impairment in QoL was found in 99% of the patients. The mean global QoL score was 6.3 ± 3.4. The impairment in QoL was positively correlated with clinical severity (r = 0.40; p = 0.003).
Conclusion: The study revealed a predominance of mild-to-moderate forms of acne, an alteration in the QoL of almost all patients studied, and a positive correlation between the clinical severity of acne and the impairment in QoL.
Key words: Acne; Clinical severity; Quality of life; Treatment; Cameroon; Sub-Saharan Africa
BACKGROUND
Acne is a chronic inflammatory pathology of the pilosebaceous follicle that progresses in flare-ups [1]. It is a visible dermatological condition [2]. It usually begins at puberty and affects between 40% and 80% of adolescents and may persist beyond adolescence or, to a lesser extent, appear in adulthood [3]. In Cameroon, Kouotou et al. found a prevalence of acne of 74.8% among educated adolescents in 2015 [4].
There are various clinical presentations of acne, among which are retentional lesions (consisting of microcysts and open comedones), superficial and deep inflammatory lesions, and scar lesions. The polymorphic clinical expression may be severe and may bear a negative impact on the quality of life (QoL) of patients and lead to psychiatric consequences, in particular, anxiety, depression, and even suicide, as demonstrated in a metanalysis by Samuels et al. in 2020 [5]. In fact, these lesions appear in adolescence, when self-esteem begins to emerge, which may affect self-confidence and the quality of relationships with others [5–7].
In Cameroon, several years ago, Kouotou et al. found a negative impact of acne on the QoL of patients as well as an association of acne with anxiety and depression [7]. The aim of this study was to evaluate the severity of acne and to assess its impact on the QoL of patients already under treatment in the Cameroonian context, a country in sub-Saharan Africa.
MATERIALS AND METHODS
Type, Place, and Duration of the Study
This was a four-month (from January to April 2017), descriptive and analytical, cross-sectional study conducted at three hospitals in Yaoundé, Cameroon. These hospitals were chosen by convenience based on the availability of a dermatologist: Biyem-Assi District Hospital (HDBA), Elig-Essono District Medical Center (CMA-EE), and Yaoundé Hospital and University Center (CHUY).
Study Population and Selection Criteria
We included patients with acne seen at the aforementioned hospitals: 1) presenting for a follow-up consultation after a medical prescription for an anti-acne medication initiated at least one month earlier; 2) received in dermatology consultation after failure of a current anti-acne treatment or withdrawn less than a month earlier; and 3) having given their informed consent to participate in the study.
Sampling
Sampling was consecutive and exhaustive during the study period. The minimum sample size was calculated with the Cochrane formula for descriptive studies considering a prevalence of 7.7% of acne reported at Cameroonian hospitals by Bissek et al. [8] and a degree of precision of 0.05, hence a minimum number of 109 participants.
Procedures
Data Collection
Data collection was performed with a technical sheet and took place in several stages.
-
1) Recruitment of participants: During dermatological consultations, the patients fulfilling the selection criteria were selected for the study.
-
2) Clinical examination: The questionnaire searched for sociodemographic characteristics, acne history and its treatment, and treatment costs. A physical examination allowed the description and classification of the clinical type and the assessment of severity with the acne lesion rating scale (ECLA) grid.
-
3) Evaluation of the impact of acne on QoL with a Cardiff Acne Disability Index (CADI) questionnaire.
Data Collection Tools
Technical Form
The technical form included sociodemographic characteristics (age, sex, profession, level of education), the history of acne, the clinical type, and the ECLA grid with its three factors (F1, F2, and F3) [9].
CADI Questionnaire (Quality-of-Life Assessment)
Employed was the CADI questionnaire: a QoL grid designed to assess QoL in adolescents and adults with acne containing a total of five items [10].
Interpretation of Scales
Interpretation of the Acne Lesion Rating Scale
The total score was obtained by adding the scores of the factors F1, F2, and F3, ranging from 0 to 36. An ECLA score less than or equal to 12 represented mild-to-moderate acne, while a score greater than 12 represented severe acne.
Interpretation of the CADI Quality-of-Life Score
The score for each response ranged from 0 to 3 points for each question. The CADI score was calculated by adding the score for each question, varying from 0 to 15. The interpretation was as follows: 1) 0: no alteration in QoL; 2) 1–5: mild alteration in QoL; 3) 6–10: moderate alteration in QoL; 4) 11–15: severe alteration QoL.
Statistical Analysis
The data collected was analyzed with SPSS, version 23.0, and Microsoft Excel 2013. The results of our study were presented in the form of tables and figures and expressed, on one hand, in terms of means ± standard deviations for the quantitative variables and, on the other, numbers and percentages for the qualitative variables. Simple linear regression was employed to assess the relationship between the ECLA score and the CADI score. We employed the ANOVA test to search for associations between sociodemographic factors and the CADI score. A p value < 0.05 was considered statistically significant.
ETHICAL CONSIDERATIONS
We obtained ethical clearance from the Institutional Ethics and Research Committee of the Faculty of Medicine and Biomedical Sciences. Research authorizations were obtained from the competent authorities of the hospitals. We conducted our study in strict accordance with the fundamental principles of the Declaration of Helsinki on research involving human subjects.
Thus, the patients and their parents or guardians for minors were informed about the various aspects of the study, the objective and the benefit, in particular, therapeutic education and the awareness of nursing staff on the psychological impact of acne. Their signed informed consent was obtained. We ensured the anonymity of the participants and the confidentiality of the information collected, which was used only for scientific purposes. Our study presented no risk to the participants.
RESULTS
Sociodemographic Characteristics
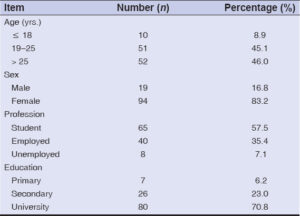
During the study period, 1930 patients were seen in dermatology consultation. Among these, we recruited 113 participants, predominantly female (83.2%), with a sex ratio of 0.2. The mean age was 26.0 ± 6.4 years, with extremes of 8 and 48 years.
Students were the most represented (57.5%) in our sample. We also noted a predominance of the higher school education level (70.8%) (Table 1).
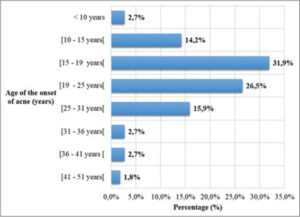
Acne most often began between the ages of fifteen and eighteen (36/113; 31.9%). In addition, in 7.2% of the cases (8/113), acne began after the age of thirty (Fig. 1).
HISTORY OF ACNE IN PATIENTS ON TREATMENT
Duration of Treatment
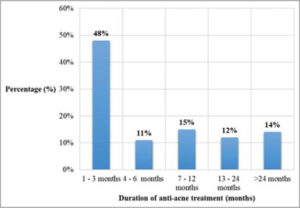
During the study, 48% of the patients had been treated for one to three months (Fig. 2).
Types of Treatments and Side Effects
In our sample, 44/113 (38.9%), 15/113 (13.3%), and 54/113 (47.8%), respectively, had undergone local treatment, oral treatment, and a combination of oral and local treatments.
Local Treatment
The most often prescribed drugs were retinoids (27.9%), followed by antibiotics (23.3%) and glycolic acid + retinaldehyde (12.8%). The most common side effects were exacerbation of acne (51.6%), followed by skin dryness and irritation (45.2%).
Oral Treatment
The most prescribed drugs were oral antibiotics (78.6%), followed by isotretinoin (20.0%). Side effects were reported in fifteen patients (13.3%): the most common were dry lips and mouth in ten out of the fourteen patients receiving isotretinoin (71.4%), as well as the exacerbation of acne (20%).
Previous Treatments
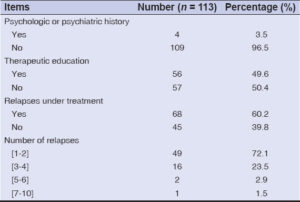
Among the 113 patients, 82 (72.6%), 23 (20.4%), and 8 (7.1%), respectively, had already experienced 1 to 2 cures, 3 to 4 cures, and at least 5 cures of treatments prior to the inclusion in our study.
Cost Evaluation
The cost of acne treatment was considered highly expensive by the majority of the patients (78/113; 69%) and affordable by 31%.
Assessment of Drug Availability
Systemic drugs remained available for the majority of the patients (79/113; 69.9%) unlike topical drugs (40/113; 35.4%).
Improvement on Treatment
Improvement in the lesions on treatment was noted by 90/113 patients (79.6%). In contrast, 14/113 patients (12.4%) saw no improvement, and 9/113 (8%) noted the worsening of their lesions (Table 2).
Identification of Clinical Forms and Assessment of Acne Severity
Papulopustular acne was the most common clinical form (54/113; 47.8%). The other clinical forms found were retentional acne (34/113; 30.1%), nodular acne (15/113; 13.3%), pigmentogenic acne (5/113; 4.4%), and acne fulminans (1/113; 0.9%). We found post-inflammatory scars in 4/113 (3.5%) patients.
The clinical assessment of severity with the ECLA score produced an average of 10.1 ± 4.6. In sum, a majority of the participants (84/113; 74.3%) had mild-to-moderate acne, while 25.7% (29/113) had a severe form.
QUALITY OF LIFE OF PATIENT WITH ACNE ON TREATMENT AND ASSOCIATED FACTORS
Quality of Life
The mean overall QoL score was 6.3 ± 3.4, with extremes of 0 and 15. In our sample, we noted a mild, moderate, and severe alteration in QoL, respectively, in 45/113 (39.8%), 54/113 (47.8%), and 13/113 (11.5%). It should be noted that QoL was not altered in one patient (0.9%).
Quality of Life and Sociodemographic Factors
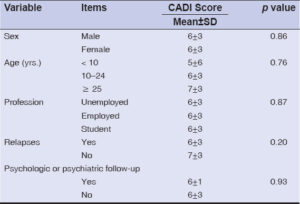
No statistically significant relationship was found between the sociodemographic variables (sex, age, profession, level of education) and the QoL score (p ≥ 0.05) (Table 3).
Quality of Life and Clinical History
Likewise, no statistically significant relationship was found between a patient’s history (relapses, psychological or psychiatric follow-ups) and the QoL score (p ≥ 0.05) (Table 3).
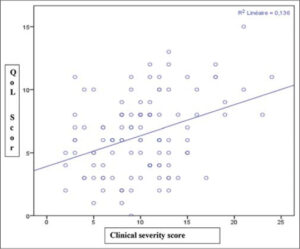
In addition, the QoL score was positively and weakly correlated with the clinical severity score (r = 0.04; p < 0.05). Indeed, the more severe was the acne, the more the QoL was altered (Fig. 3).
DISCUSSION
Our study focused on the clinical severity of acne on black skin and assessed the impact of acne on the QoL of patients already on anti-acne treatment in the Cameroonian context, a country in sub-Saharan Africa.
In our series, acne mainly began between the ages of fifteen and eighteen [11,12]. In fact, based on the pathophysiological aspect, acne began at the age of puberty with the androgenic hormonal secretions that it causes. However, in a significant proportion of our participants (7.2%), acne began after the age of thirty. A late onset of acne is not uncommon; it is often the consequence of a genetic predisposition, hyperandrogenism, or the use of depigmenting cosmetics [13]. The mean age was 26.0 ± 6.4 years, with extremes of 8 and 48 years. Similar results were noted two years earlier by Kouotou et al. in Cameroon (25.4 ± 7.1 years) [7]. This observation was also made in other studies such as that by Saka et al. in Togo (23.7 ± 5.7 years) [14] and Dégboé et al. in Benin (24.6 ± 8.5 years) [15]. Acne usually begins in the early teens around the age of twelve, yet the first dermatological consultation is almost always done too late. This delay in consultation observed in our context could be explained by social considerations. Indeed, acne is often overlooked and considered transient in adolescents in our community. Females were more affected (83.2%) by acne in our series. The female predominance was corroborated by numerous series in the literature [7,14,15]. The high proportion of females consulting for acne is linked to the readiness of females to consult more easily than males, especially in the case of skin diseases.
In our patients, the duration of treatment varied from one month to five years. The duration of five years could be explained by inadequate self-medication or poor access to anti-acne drugs [16]. On clinical examination, papulopustular acne was the major clinical form (86.7%) and the clinical severity was classified as mild-to-moderate in 74% of the participants. Other studies also noted the predominance of mild-to-moderate forms on both white and black skin [7,11,14,15]. The anti-acne treatment initiated was most often topical (86.7%), sometimes combined with oral treatment (47.8%), which was in accordance with the acne management algorithm [17]. Due to the chronicity of acne lesions, follow-up is long and lasts several months or even years, requiring frequent reassessments. It usually takes several months to receive a satisfactory result for each treatment initiated [18]. In our case, 48% of the patients had already been on treatment for one to three months and 14% of them had been treated and followed for more than twenty-four months.
The unsightly and chronic nature of acne as well as the long duration of treatment have an impact on the QoL of the patient suffering from acne. QoL was assessed during our work with the Cardiff Acne Disability Index (CADI), an assessment grid for the QoL of patients with acne. QoL was altered in 99% of the participants. The alteration in QoL was mild, moderate, and severe in 40%, 48%, and 11% of the participants, respectively. Around two years earlier, Kouotou et al. found almost identical results in the same city. QoL was altered in all participants; more specifically, it was slightly, moderately, and severely impaired in 49.2%, 41.4%, and 9.4% of patients, respectively [5]. Likewise, in 2016, El-Hamd et al. found trends similar to ours in their study in Egypt, notably, with a mild (54%), moderate (33%), and severe (13%) alteration in the QoL of their patients with acne [19]. Overall, acne patients almost always experience an altered QoL. It would be important to systematically assess the QoL of patients with acne in order to take this into account during management. In addition, some quasi-experimental studies increasingly assess the QoL of the patient before and after treatment, in addition to evaluating the clinical efficacy of the anti-acne treatment [20,21]. The CADI tool could also serve as a routine tool for assessing improvement in QoL during initial and follow-up consultations and help to improve patient care.
Additionally, we noted a significant positive linear correlation between the ECLA score and the CADI score (r = 0.04; p < 0.05). Numerous African studies showed an association between the severity of clinical lesions and an impaired QoL [7,19,21]. The unsightly or even frightening aspect of severe acne lesions, such as nodulocysts, and the profuse nature of moderate-to-severe acne lesions may draw the attention of others to question and pity the patient, thus having a more negative impact on their QoL. In contrast, the team of Gupta et al. did not find a linear correlation between the clinical severity of lesions and the CADI score (p > 0.05). In a society seeking beauty and well-being increasingly often, it is possible that QoL is impaired regardless of the severity of clinical lesions [11]. In addition, there was no statistically significant difference in the CADI score depending on sex, profession, relapses, and psychological follow-up in our participants (p > 0.05). These sociodemographic factors and the clinical history did not influence the QoL of our patients. This trend was also underlined by Cameroonian [7] and Indian [11] studies. However, environmental factors, in particular, tobacco and alcohol consumption, may have worsened the CADI score of the patients, as evidenced by the work of Gupta et al. [11]. Analytical studies evaluating the effect of environmental factors on the QoL of patients with acne could be performed to document this finding and improve the QoL of these patients.
Limitations of the Study
Certain aspects of this work could constitute limits. The data collection tools, namely the ECLA and CADI grids, which assessed QoL, were validated against a Caucasian population, not Negroid. Nevertheless, our study could constitute a preliminary for further work with rating scales more suitable for dark skin.
CONCLUSION
Our study allowed us to discover that papulopustular acne was the most common clinical form and that acne in our participants was generally mild-to-moderate according to the ECLA scoring grid. Acne almost always induced an altered QoL in the patients. This alteration in QoL was significantly correlated with the clinical severity of lesions. Sociodemographic factors did not influence the QoL of our participants. The assessment of QoL in current practice in front of any patient suffering from acne may help to further understand the patient’s suffering with acne and improve its management.
Statement of Human and Animal Rights
All the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the 2008 revision of the Declaration of Helsinki of 1975.
Statement of Informed Consent
Informed consent for participation in this study was obtained from all patients.
REFERENCES
1. Muguet Guenot L, Vourc’h Jourdain M, Saint Jean M, Corvec S, Lemoigne M, Khammari A, et al. Microscopie confocale et acnéde la femme adulte. Ann Dermatol Vénéréol. 2017;144(12, Supplement):S177-8.
2. Troin L, Mallet S, Lagouanelle M-C, Scannapieco F, Lignon C, Gaudy-Marqueste C, et al. [Five years’experience of cosmetic camouflage of disfiguring skin disorders:Patient satisfaction]. Ann Dermatol Vénéréol. 2020;147:4-8.
3. Wolkenstein P, MachovcováA, Szepietowski JC, Tennstedt D, Veraldi S, Delarue A. Acne prevalence and associations with lifestyle:A cross-sectional online survey of adolescents/young adults in 7 European countries. J Eur Acad Dermatol Venereol. 2018;32:298-306.
4. Kouotou EA, Nansseu NJR, Defo D, Bissek ACZ-K. AcnéJuvénile :Une Pathologie Fréquente Chez Les Adolescents Scolarisés D’Afrique Sub-saharienne. Health Sci Dis. 2015;16.
5. Samuels DV, Rosenthal R, Lin R, Chaudhari S, Natsuaki MN. Acne vulgaris and risk of depression and anxiety:A meta-analytic review. JAAD. 2020;83:532-41.
6. Saka B, Noude Téclessou J, Akakpo SA, Mahamadou G, Kassang P, GnossikèP, et al. Acnéet comorbidités psychiatriques àLomé, Togo:étude cas-témoin. Ann Dermatol Vénéréol. 2019;146(12, Supplement):A205.
7. Kouotou EA, Adegbidi H, Bene Belembe R, Sieleunou I, Nansseu JR, Kamga J-P, et al. [Acne in Cameroon:Quality of life and psychiatric comorbidities]. Ann Dermatol Vénéréol. 2016;143:601-6.
8. Bissek AZ-K, Kouotou E, Defo D, Njamnshi KA, Koueke P, Muna W. Epidémiologie des dermatoses àl’Hôpital Général de Yaoundé. Health Sci Dis. 2009;10.
9. Dreno B, Bodoch I, Chivot M, Daniel F, Humbert P, POLI F, et al. La grille ECLA :un système de cotation de l’acnépour la pratique quotidienne du dermatologue. Ann Dermatol Vénéréol. Paris:Masson;1999;126:136-41.
10. Motley RJ, Finlay AY. Practical use of a disability index in the routine management of acne. CED. 1992;17:1-3.
11. Gupta A, Sharma Y, Dash K, Chaudhari N, Jethani S. Quality of life in acne vulgaris:Relationship to clinical severity and demographic data. Int J Dermatol. 2016;82:292.
12. Ouedraogo AN, Kabre Ouedraogo SSS, Ouedraogo MS, Traore F, Tapsoba PG, Barro-Traore F, et al. Acne:Prevalence, perceptions and beliefs among pupils and students in Ouagadougou, Burkina Faso. Our Dermatol Online. 2017;8:10-6.
13. Poli F, Faye O, Ly F, Le Thuaut A. [Acne in adult female patients:A comparative study in France and sub-Saharan Africa]. Ann Dermatol Vénéréol. 2014;141:336-45.
14. Saka B, Akakpo AS, Téclessou JN, Mouhari-Toure A, Mahamadou G, Gnossike P, et al. Acne in Lomé, Togo:Clinical aspects and quality of life of patients. BMC Dermatol. 2018;18:7.
15. DégboéB, Koudoukpo C, Agbéssi N, Elégbédé-AdégbitèN, Akpadjan F, Adégbidi H, et al. Acne on pigmented skin:Epidemiological, clinical and therapeutic features in dermatology in Benin. JCDSA. 2019;09:305.
16. DégboéBE, Koudoukpo C, Agbéssi N, Akpadjan F, Adégbidi H, AtadokpèdéF. Acnésur peau noire:facteurs associés et comorbidités psychiatriques dans les services de dermatologie du Bénin. Ann Dermatol Vénéréol. 2019;146(12, Supplement):A205-6.
17. Le Cleach L, Lebrun-Vignes B, Bachelot A, Beer F, Berger P, Brugère S, et al. Prise en charge de l’acné. Traitement de l’acnépar voie locale et générale. Ann Dermatol Vénéréol. 2015;142:692-700.
18. Couic-Marinier F, Pillon F, Chambin O. Renouvellement d’une ordonnance anti-acnéique. Actual Pharm. 2014;53:11-3.
19. El-Hamd MA, Nada EE-DA, Moustafa MA-K, Mahboob-Allah RA. Prevalence of acne vulgaris and its impact of the quality of life among secondary school-aged adolescents in Sohag Province, Upper Egypt. JCD. 2017;16:370-3.
20. Cyrulnik AA, Viola KV, Gewirtzman AJ, Cohen SR. High-dose isotretinoin in acne vulgaris:Improved treatment outcomes and quality of life. Int J Dermatol. 2012;51:1123-30.
21. Zaraa I, Belghith I, Ben Alaya N, Trojjet S, Mokni M, Ben Osman A. Severity of acne and its impact on quality of life. Skinmed. 2013;11:148-53.
Notes
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0000-0003-3879-2659 http://orcid.org/0000-0003-3879-2659 http://orcid.org/0000-0003-0630-2678 http://orcid.org/0000-0003-0630-2678 |




Comments are closed.