A study on the association between thyroid dysfunction and various dermatoses in northeastern India
Nandakishore Thokchom1, Kapila Verma2, Bishurul NA Hafi3, Sandhyarani Kshetrimayum4, Vanlalhriatpuii Hmar5, Linda Kongbam 1, Nandita Bhattacharjee6
1, Nandita Bhattacharjee6
1Department of Dermatology, Venereology and Leprology, Regional Institute of Medical Sciences, Imphal, Manipur, India, 2Department of Dermatology, Venereology and Leprology, LN Medical College and JK Hospital, Bhopal, Madhya Pradesh, India, 3IQRRA International Hospital and Research Centre, Kozhikode, Kerala, India, 4Serenity Skin Hair and Medicine Clinic, Bengaluru, Karnataka, India, 5Medical Officer, Government of Mizoram, Mizoram, India, 6Indira Gandhi Memorial Hospital, Agartala, Tripura, India
Corresponding author: Linda Kongbam, MD
How to cite this article: Thokchom N, Verma K, Hafi BNA, Kshetrimayum S, Hmar V, Kongbam L, Bhattacharjee N. A study on the association between thyroid dysfunction and various dermatoses in northeastern India. Our Dermatol Online. 2022;13(2):143-147.
Submission: 15.08.2021; Acceptance: 19.11.2021
DOI: 10.7241/ourd.20222.5
Citation tools:
Copyright information
© Our Dermatology Online 2022. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
ABSTRACT
Background: Thyroid abnormalities are a well-documented endocrine association seen in numerous dermatological conditions. However, there are few Indian studies analyzing the thyroid status in various dermatoses.
Materials and Methods: This cross-sectional, observational study was conducted on 805 willing patients with various dermatoses, irrespective of the disease classification. Serum samples were taken and assayed for triiodothyronine (T3), thyroxine (T4), and thyroid-stimulating hormone (TSH).
Results: Out of the 805 patients, 14.2% (n = 115) had abnormal thyroid function. Hypothyroidism accounted for 86.1% (n = 99) and hyperthyroidism for 13.9% (n = 16). Among the 99 hypothyroid patients, 23.2% (n = 23) had overt hypothyroidism while 76.8% (n = 76) had subclinical hypothyroidism. Vitiligo accounted for 36.1% (n = 291) of the patients, among which 12.3% (n = 36) had thyroid dysfunction involving six hypothyroid and three hyperthyroid cases. Out of 58 cases of melasma, 15 (25.8%) had abnormal thyroid function. Among the twenty patients with lichen planus pigmentosus, five had abnormal thyroid levels.
Conclusion: This study demonstrates a strong association of thyroid disorders in certain conditions, such as vitiligo, alopecia areata, diffuse hair loss, dry skin, chronic urticaria, and melasma. Thus, the inclusion of thyroid function tests in routine investigations of these conditions would be helpful.
Key words: hypothyroidism; hyperthyroidism; dermatoses; vitiligo; melasma; lichen planus pigmentosus
INTRODUCTION
Endocrine disorders are associated with various skin manifestations, specific or non-specific. Identifying the underlying endocrinopathy allows for early diagnosis and appropriate treatment of the patient rather than symptomatic treatment. Thyroid disorders are important endocrine diseases having cutaneous manifestations, as the skin and its appendages are target organs of thyroid hormones [1]. Although most of these cutaneous changes are non-specific, a proper clinical evaluation with a laboratory confirmation may aid in the diagnosis in the early stage [2]. The most frequent thyroid dysfunction in India is hypothyroidism [3]. Some studies have been done in India assessing thyroid function in certain dermatologic conditions, such as vitiligo, melasma, and polymorphic light eruptions, yet studies on a wider range of dermatologic diseases are missing [4–6]. Therefore, the present study was conducted to assess the association of thyroid dysfunction with various dermatoses in northeastern India.
MATERIALS AND METHODS
A cross-sectional study was conducted on 805 patients presenting with dermatoses, irrespective of the disease classification, to the dermatology OPD of a tertiary center in northeastern India for a period of three years (April 2013 to May 2016). Pregnant females, individuals with known endocrine dysfunction, and those who refused to consent were excluded. Detailed clinical history taking and a physical examination were performed. Serum samples were collected and assayed for serum triiodothyronine (T3), thyroxine (T4), and thyroid-stimulating hormone (TSH). Thyroid hormones were assayed by the VIDAS automated quantitative test with an enzyme-linked fluorescent assay. The reference ranges for serum T3, T4, and TSH were 0.9–2.5 nmol/L, 60–120 nmol/L, and 0.3–5 IU/mL, respectively. The diagnosis of hypothyroidism was established when thyroid function exhibited raised TSH levels with or without low T3 and T4 levels. Subclinical hypothyroidism is defined by normal serum T3 and T4 levels but TSH > 5 IU/mL and TSH < 15 IU/mL. Hyperthyroidism was diagnosed if serum T3 and T4 were raised and TSH was low. Subclinical hyperthyroidism was diagnosed if the TSH level was undetectable and with serum T3 and T4 in the upper limit of the reference range. Ethical clearance was obtained from the institute ethical committee. Data was tabulated and analyzed with SPSS, version 21.
RESULTS
Among 805 patients with various skin disorders, 199 were males (24.7 %) and 606 were females (75.3%). The youngest patient was four months old and the oldest was 84 years old. Most were in the age group 11–20 years (Fig. 1). Abnormal thyroid function was observed in 14.2% (n = 115) of the patients (Fig. 2). Hypothyroidism accounted for 86.1% (n = 99) and hyperthyroidism for 13.9% (n = 16) of the cases (Fig. 2). Vitiligo accounted for 36.1% (n = 291) of the cases, among which 12.3% (n = 36) had thyroid dysfunction involving nine overt thyroid dysfunction cases (six hypothyroid and three hyperthyroid cases). Among 58 cases of melasma, 15 had abnormal thyroid function. Among 20 patients suffering from lichen planus pigmentosus (LPP), five patients had abnormal thyroid levels. Among hair disorders, alopecia areata (AA) and diffuse hair loss had nine and seven abnormal thyroid values, respectively. Among 114 cases of urticaria, 9 (7.9%) had thyroid dysfunction. Table 1 shows various other cutaneous disorders with their respective thyroid levels. Among 99 hypothyroid patients, 23.2% (n = 23) had overt hypothyroidism, whereas 76.8% (n = 76) revealed subclinical hypothyroidism (Fig. 2). Table 2 lists the dermatoses associated with overt hypothyroidism or hyperthyroidism.
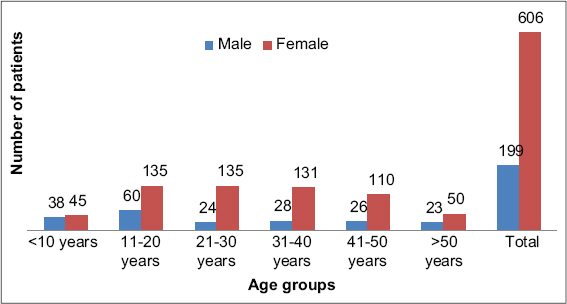 |
Figure 1: Age and gender distribution. |
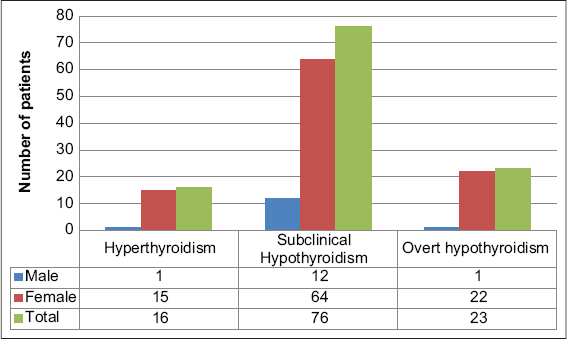 |
Figure 2: Distribution of thyroid disorders. |
 |
Table 1: Thyroid status in various dermatoses |
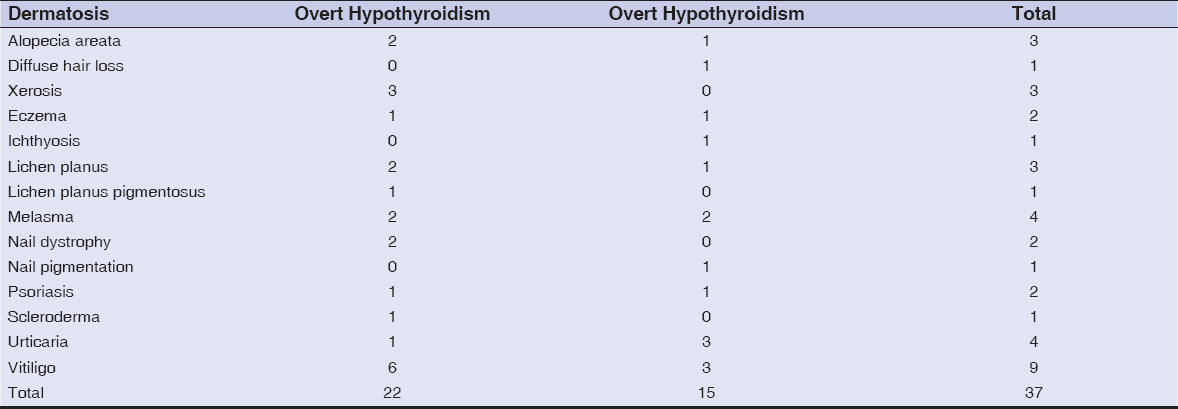 |
Table 2: Dermatoses with overt hypothyroidism and hyperthyroidism |
DISCUSSION
The present study documented thyroid dysfunction in 12.3% of vitiligo patients. A similar association (12%) was noted by another study from northern India [7]. However, a recent Indian study has shown a higher proportion (28%) of thyroid dysfunction and higher antithyroid antibodies when compared to controls [8]. In concordance with the findings from different parts of India, this study also reveals a high prevalence of thyroid dysfunction with predominant hypothyroidism among vitiligo patients.
A high incidence of thyroid dysfunction among melasma patients was reported by various studies earlier. An earlier study revealed that the frequency of thyroid disorders was higher in melisma patients than in controls [9]. In another study, 26.2% of melasma patients had hypothyroidism [10]. In the present study, 25.9% of melasma cases showed thyroid dysfunction. However, there are few controlled studies on melasma and thyroid disorders [9,11]. Therefore, more studies are required to determine the association between melasma and thyroid disorders. Both melasma and thyroid diseases are highly common in young females, hence this may lead to the incidental finding of thyroid dysfunction in melasma patients [12].
In the present study, 25% of LPP patients had hypothyroidism, which is comparable to a study by Karn et al. in which 31.7% of patients with LPP had hypothyroidism and three had hyperthyroidism. Studies with a larger number of patients are required to substantiate a significant association between the two conditions [13].
Among the 60 patients with AA in this study, nine had thyroid dysfunction, among which eight were hypothyroidism cases and one was a hyperthyroidism case. Earlier studies revealed a significant association between AA and thyroid disorders [14,15]. Similar findings were also observed in a study by Ijas et al. [16]. All these findings suggest that, for patients with AA, testing for thyroid function should be mandatory. Other authors have reported a similar association [17–19].
Premature graying of the hair, or canities, is a common hair condition of unknown etiology. In this study, only three cases were evaluated and all had normal thyroid function. A study by Daulatabad et al. also failed to identify any significant correlation between canities and thyroid status [20].
A significant association between chronic urticaria and thyroid disorders was reported in previous studies [21–24]. In this study also, 7.9% of the patients with chronic spontaneous urticaria exhibited thyroid dysfunction.
Among the 14 psoriatic patients, four revealed abnormal thyroid function in this study. However, other studies reported no significant association [25–27].
Other dermatological conditions, especially of autoimmune origin, have also been reported to have associated thyroid disorders, such as morphea, lupus erythematosus, scleroderma, dermatitis herpetiformis, erythema annulare centrifugum, generalized granuloma annulare, reticular erythematous mucinosis, pseudoxanthoma elasticum, and palmoplantar pustulosis [28].
Cutaneous manifestations may serve as an early indicator of thyroid disorders. Also, a high prevalence of subclinical hypothyroidism in many patients with various cutaneous disorders may indicate that subclinical hypothyroidism may be an important unrecognized association with numerous cutaneous disorders. Subclinical hypothyroidism was the most prevalent thyroid gland disease in one study [29].
The limitation of the present study was that tests such as antithyroid antibodies or a radioactive thyroid scan were not performed to determine the etiological nature of thyroid dysfunction in the cases of hypothyroidism and hyperthyroidism. Moreover, as all types of dermatological disorders were included in the study, it was impractical to find an equally matched control group from an outpatient set-up to be able to conduct a case-control study.
CONCLUSION
Thyroid disorders are often observed in numerous dermatological conditions, such as vitiligo, alopecia areata, diffuse hair loss, dry skin, chronic urticaria, and melasma. This suggests the inclusion of thyroid function tests in the routine investigations of these conditions. Cutaneous manifestations may also be the only symptom or the earliest symptom of thyroid disorders. Therefore, a periodic assessment of thyroid function is necessary for the early diagnosis and management of the underlying thyroid dysfunction.
Statement of Human and Animal Rights
All the procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the 2008 revision of the Declaration of Helsinki of 1975.
Statement of Informed Consent
Informed consent for participation in this study was obtained from all patients.
REFERENCES
1. Safer JD. Thyroid hormone action on skin. Dermatoendocrinol. 2011;3:211-5.
2. Puri N. A study on cutaneous manifestations of thyroid disease. Indian J Dermatol. 2012;57:247.
3. Deokar PG, Nagdeote AN, Lanje MJ, Basutkar DG. Prevalence of thyroid disorders in a tertiary care center. Int J Curr Res Rev. 2016;8:26.
4. Achar A, Rathi SK. Melasma:a clinico-epidemiological study of 312 cases. Indian J Dermatol. 2011;56:380-2.
5. Gopal K, Rao GR, Kumar YH. Increased prevalence of thyroid dysfunction and diabetes mellitus in Indian vitiligo patients:A case-control study. Indian Dermatol Online J. 2014;5:456-60.
6. Sharma L, Lamba S, Singh SK. Thyroid function tests in cases of polymorphic light eruption:A case-control study. Indian Dermatol Online J. 2014;5:291-5.
7. Singh S, Usha, Pandey SS. Epidemiological profile of vitiligo in northern India. J App Pharm Sci. 2011;1:211-4.
8. Biswas M, Chattopadhyay A, Mridha K, Biswas T, Biswas J, Hassan K. A study on association between vitiligo and thyroid dysfunction. J Dent Med Sci. 2015;14:34-7.
9. Rostami Mogaddam M, Iranparvar Alamdari M, Maleki N, Safavi Ardabili N, Abedkouhi S. Evaluation of autoimmune thyroid disease in melasma. J Cosmet Dermatol. 2015;14:167-71.
10. Suthanther CB, Bubna AK, Sankarasubramanian A, Veeraraghavan M, Rangarajan S, Muralidhar K. A clinical study of melasma and assessment of Dermatology Life Quality Index at a tertiary health care center in South India. Pigment Int. 2016;3:77.
11. Abrol S, Sharma R. A clinicoepidemiological study of facial hypermelanosis among females of reproductive age group. Our Dermatol Online. 2019;10:239-43.
12. Handel AC, Miot LD, Miot HA. Melasma:A clinical and epidemiological review. An Bras Dermatol. 2014;89:771-82.
13. Karn D, Kc S, Timalsina M. Lichen planus pigmentosus:A study for association of thyroid dysfunction. Kathmandu Univ Med J. 2016;14:36-40.
14. Kouassi YI, Gbandama KKP, Kourouma HS, Kouassi KA, Allou A-S, Kaloga M, et al. Alopecia areata in Black African patients:Epidemiological, clinical, and therapeutic aspects. Our Dermatol Online. 2021;12:24-6.
15. Lee S, Lee YB, Kim BJ, Lee WS. Screening of thyroid function and autoantibodies in patients with alopecia areata:A systematic review and meta-analysis. J Am Acad Dermatol. 2019;80:1410-3.
16. Ahmed I, Nasreen S, Jehangir U, Wahid Z. Clinical spectrum of alopecia areata and its association with thyroid dysfunction. J Pak Assoc Dermatol. 2012;22:207-12.
17. Shashikant M. A hospital-based cross-sectional study to determine causes of diffuse hair loss in women. J Clin Diagn Res. 2015;9:1-4.
18. Jayashankar CA, Shailaja A, Prakash B, Swetha HP. Hemoglobin, ferritin and thyroid profile in women with chronic telogen effluvium. Int J Res Med Sci. 2016;4:152-5.
19. Shaikh L, Almulhim A, Al Rabai M, Shaikh Y. Effective treatment of alopecia universalis with oral tofacitinib:A case report. Our Dermatol Online. 2021;12:33-6.
20. Daulatabad D, Singal A, Grover C, Chhillar N. Profile of Indian patients with premature canities. Indian J Dermatol Venereol Leprol. 2016;82:169-72.
21. Yadav S, Kanwar AJ, Prasad D, Minz RW. Chronic idiopathic urticaria and thyroid autoimmunity:Perplexing association. Indian J Dermatol. 2013;58:325-9.
22. Pan XF, Gu JQ, Shan ZY. The prevalence of thyroid autoimmunity in patients with urticaria:A systematic review and meta-analysis. Endocrine. 2015;48:804-10.
23. Keen MA, Shah FY. Oral mini-pulse steroid therapy in severe chronic urticaria. Our Dermatol Online. 2021;12:e83.
24. Nuzzo V, Tauchmanova L, Colasanti P, Zuccoli A, Cola A. Idiopathic chronic urticaria and thyroid autoimmunity:Experience of a single centre. Dermatoendocrinology 2011;4:255-8.
25. Robati RM, Toossi P, Roodsari MR, Khalilazar S, Abolhasani E, Namazi N, et al. Association of psoriasis severity with serum prolactin, thyroid hormones, and cortisol before and after treatment. ScientificWorldJournal. 2013;3:1-5.
26. Namiki K, Kamata M, Shimizu T, Chijiwa C, Uchida H, Okinaga S, et al. Thyroid dysfunction in patients with psoriasis:Higher prevalence of thyroid dysfunction in patients with generalized pustular psoriasis. J Dermatol. 2020;47:133-9.
27. Lai YC, Yew YW. Psoriasis and thyroid profile:Analysis of the U.S. National Health and Nutrition Examination Survey database. Indian J Dermatol Venereol Leprol. 2016;82:310-2.
28. Sijapati KS, Rijal A, Agrawal S, Khadka DK, Maskey R. Cutaneous manifestations of thyroid hormone disorder. Nep J Dermatol Venereol Leprol. 2019;17:42-8.
29. Sulejmanovic M, Cickusic AJ, Salkic S, Bousbija FM. Annual incidence of thyroid disease in patients who first time visit department for thyroid diseases in Tuzla Canton. Mater Sociomed. 2019;31:130-4.
Notes
Source of Support: Nil,
Conflict of Interest:
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/0000-0001-9749-8906 http://orcid.org/0000-0001-9749-8906 |



Comments are closed.