Symmetric periorbital edema after mRNA COVID-19 vaccine
Veselina Belyova1, Razvigor Darlenski 1,2
1,2
1Department of Dermatology and Venereology, Acibadem City Clinic Tokuda Hospital, Sofia, Bulgaria, 2Department of Dermatology and Venereology, Acibadem City Clinic Tokuda Hospital, Sofia, Bulgaria, Section of Dermatovenereology, Trakia University, Stara Zagora, Bulgaria
Corresponding author: Prof. Razvigor Darlenski, MD PhD
How to cite this article: Belyova V, Darlenski R. Symmetric periorbital edema after mRNA COVID-19 vaccine. Our Dermatol Online. 2022;13(2):210.
Submission: 14.01.2022; Acceptance: 03.02.2022
DOI: 10.7241/ourd.20222.22
Citation tools:
Copyright information
© Our Dermatology Online 2022. No commercial re-use. See rights and permissions. Published by Our Dermatology Online.
A 42-year-old Caucasian female patient presented with symmetric, asymptomatic, periorbital angioedema (Fig. 1) that appeared 24 hours after the second dose of the Moderna mRNA COVID-19 vaccine. She was injected with the hyaluronic acid dermal filler Juvéderm Volite in the glabella and periorbital area three months earlier and underwent a botulinum toxin session in the forehead, glabella, and crow’s feet with Vistabel six months earlier. No present or previous history of allergy or concomitant diseases was reported. Cetirizine dihydrochloride 3 × 10 mg and cold compresses resulted in the complete resolution of the symptoms within three days.
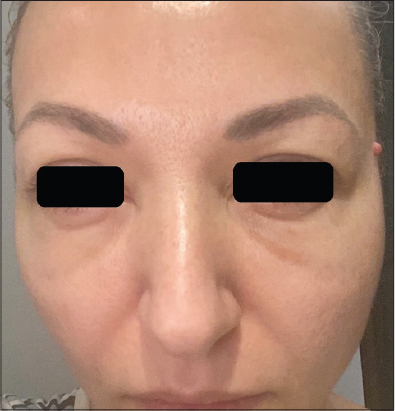 |
Figure 1: Symmetric periorbital edema developed 24 hours after the second dose of the Moderna mRNA COVID-19 vaccine. |
Reactions to the Moderna vaccine possibly related to the dermal filler placement are rare and have an unknown pathogenesis. Three out of 15,184 patients developed facial or lip swelling after mRNA vaccine [1]. These reactions may be immunologically triggered by viral or bacterial illness and vaccinations, including allergies to vaccine ingredients such as polyethylene glycol [2].
ACKNOWLEDGMENTS
The patient described by this article gave written informed consent to the publication of their case details.
Consent
The examination of the patient was conducted according to the principles of the Declaration of Helsinki.
The authors certify that they have obtained all appropriate patient consent forms, in which the patients gave their consent for images and other clinical information to be included in the journal. The patients understand that their names and initials will not be published and due effort will be made to conceal their identity, but that anonymity cannot be guaranteed.
REFERENCES
1. Avram M, Bertucci V, Cox SE, Jones D, Mariwalla K. Guidance Regarding SARS-CoV-2 mRNA Vaccine Side Effects in Dermal Filler Patients. American Society for Dermatologic Surgery;December 28, 2020. Accessed March 22, 2021. https://www.asds.net/Portals/0/PDF/secure/ASDS-SARS-CoV-2-Vaccine-Guidance.pdf
2. Banerji A, Weckner P, Saff R, Stone Jr C, Robinson L. mRNA Vaccines to prevent COVID-19 disease and reported allergic reactions:Current evidence and suggested approach. J Allergy Clin Immunol Pract. 2021;9:1423-37.
Notes
Source of Support: Nil,
Conflict of Interest: None declared.
Request permissions
If you wish to reuse any or all of this article please use the e-mail (brzezoo77@yahoo.com) to contact with publisher.
| Related Articles | Search Authors in |
|
 http://orcid.org/000-0002-4220-8040 http://orcid.org/000-0002-4220-8040 |



Comments are closed.