|
Get Citation
|
|
|
Jędrowiak A, Kobusiewicz A, Trznadel-Grodzka E, Kaszuba A. Dermoscopic fi ndings in extragenital lichen sclerosus. Our Dermatol Online. 2018;9(2):197-199. |
|
|
Download citation file:
|
Dermoscopic findings in extragenital lichen sclerosus
Anna Jędrowiak, Aleksandra Kobusiewicz, Ewa Trznadel-Grodzka, Andrzej Kaszuba
Department of Dermatology, Pediatric Dermatology and Dermatologic Oncology, Medical University of Lodz, Lodz, Poland
Corresponding author: Anna Jędrowiak, E-mail: aniaula@o2.pl
Submission: 01.08.2017; Acceptance: 27.10.2017
DOI: 10.7241/ourd.20182.24
ABSTRACT
Lichen sclerosus (LS), also known as lichen sclerosus et atrophicus is a chronic inflammatory dermatosis of unknown aetiology. It has both genital and extragenital presentations, nevertheless genital forms significantly outnumber extragenital LS. Dermoscopy is noninvasive diagnostic tool traditionally employed in pigmented lesions, however its usefulness in inflammatory skin conditions is becoming continuously more meaningful. Although the clinical diagnosis of fully developed LS rarely causes difficulties, unusual presentations require differentiation from the diseases such as lichen planus, morphea, extramammary Paget disease, SCC and others. In these cases histopathology contributes to the diagnosis. Studies on the use of dermoscopy in LS are sparse, nevertheless some dermoscopic features of LS has been described.
Key words: Extragenital lichen sclerosus, Dermoscopy, Lichen sclerosus
INTRODUCTION
Lichen sclerosus (LS)is a chronic, usually asymptomatic, inflammatory dermatosis that results in epidermal atrophy and scarring. It is prevalent in females with bimodal onset in prepubertal and postmenopausal age group [1,2]. It most often affects genital and perianal areas of postmenopausal women, however it can affect men and pre-pubertal children. LS is ten times more common in women than in men. Extragenital LS affects 10 % of women with vulval disease [3]. Penile LS is the leading cause of the phimosis in adult men [1]. Genital LS has been associated with a certain risk of squamous cell carcinoma (SCC), whereas extragenital LS does not appear to predispose to cancer. Cancer is estimated to affect up to 5 % of patients with vulval, penile or anal LS [3].
The exact aetiology of LS has not been ascertained yet, however, evidence points to an increased likelihood of autoimmune and genetic component. The most common autoimmune diseases associated with LS are autoimmune thyroiditis, alopecia areata, vitiligo and pernious anemia [3].
CASE REPORT
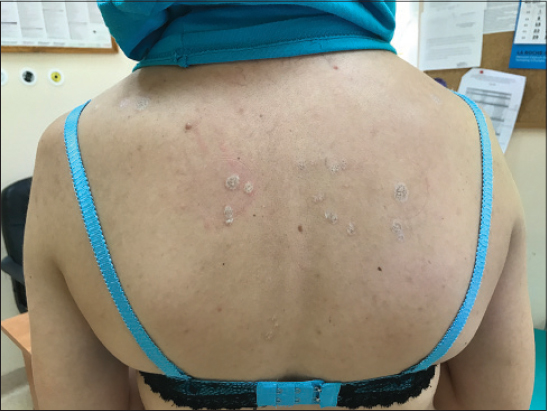
A 32-year-old woman with no previous medical history of autoimmune diseases, with multiple itchy, hypopigmented, atrophic, well-demarcated, brightening round and oval papules and plaques on the upper back since 1,5 year (Fig. 1). Hair, nails, oral mucosa and anogenital region were unaffected. Blood investigations did not show any abnormalities. The patient has not been treated yet.
DISCUSSION
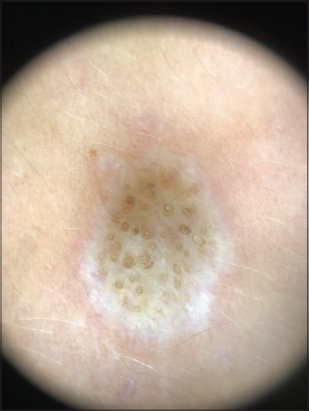
The clinical differentiation of LS and morphea, especially in extragenital regions, is a diagnostic challenge. White structureless areas and comedo-like openings are typically seen in LS [4], whereas fibrotic bands are characteristic of morphea. Nevertheless, comma shaped vessels, hairpin like vessels and dotted vessels are usually absent in morphea and are seen only in LS [5].
Comedo-like openings in LS are predominat in early lesions, whereas white chrysalis like structures suggest homogenization of collagen in the dermis and can be seen only in late lesions. Long persisting lesions appear atrophic [2,6].
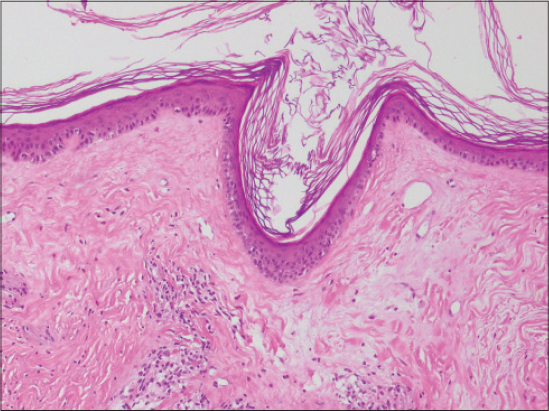
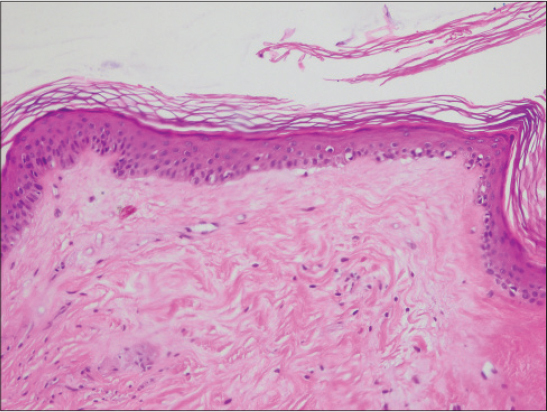
Dermoscopic structures in LS correlate with histopathology: white structureless areas are representing epidermal atrophy, whereas comedo-like openings are representing follicular plugging in histopathology [5].
Histopathology of morphea shows the continuity of the basal membrane zone (BMZ), whereas in LS numerous invaginations are present in BMZ [7].
CONCLUSION
Although the clinical diagnosis of fully developed LS rarely causes difficulties and the disease is usually recognized by its appearance, early forms or unusual presentations require differentiation from other diseases including morphea, lichen planus, SCC and others. In these cases histopathology contributes to the diagnosis and is mandatory for any clinical situation in which co-existing SCC cannot be ruled out. Early diagnosis and treatment play a substantial role in patient’s prognosis and result in decreased risk of malignancy and scarring.
REFERENCES
1. Larre Borges A, Tiodorovic-Zivkovic D, Lallas A, Moscarella E, Gurgitano S, Capurro M, et al. Clinical, dermoscopic and histopathologic features of genital and extragenital lichen sclerosus. J Eur Acad Dermatol Venereol. 2013;27:1433-9.
2. Ankad BS, Beergouder SL. Dermoscopic patterns in lichen sclerosus:A report of three cases. Indian Dermatol Online J. 2015;6:237-40.
3. Pragya Ashok Nair P. Vulvar Lichen Sclerosus et Atrophicus, J Midlife Health. 2017;8:55–62.
4. Horcajada-Reales C, Campos-Dominiquez M, Conde-Montero E, Parra-Blanco V, Suarez-Fernandez R. Comedo-like openings in dermoscopy:an essential diagnostic clue for lichen sclerosus, even in children. J Am Acad Dermatol. 2015;72(1 Suppl):S4-5.
5. Shim WH, Jwa SW, Song M, Kim HS, Ko HC, Kim MB, et al. Diagnostic usefulness of dermatoscopy in differentiating lichen sclerous et atrophicus from morphea. J Am Acad Dermatol. 2012;66:690–1.
6. Garrido-Ríos AA, Alvarez-Garrido H, Sanz-Muñoz C, Aragoneses-Fraile H, Manchado-López P, Miranda-Romero A. Dermoscopy of extragenital lichen sclerosus. Arch Dermatol. 2009;145:1468.
7. Kowalewski C, Kozlowska A, Gorska M, Wozniak K, Krajewski M, Blaszczyk M, et al. Alterations of basement membrane zone and cutaneous microvasculature in morphea and extragenital lichen sclerosus. Am J Dermatopathol. 2005;27:489–96.
Notes
Source of Support: Nil
Conflict of Interest: None declared.
Comments are closed.