|
Get Citation
|
|
|
Rafiei R, Hojat E, Daryakar A, Yusefkhani L, Rafiee B. Anetoderma with positive Darier’s sign. Our Dermatol Online. 2017;8(1):53-55. |
|
|
Download citation file:
|
Anetoderma with positive Darier’s sign
Rana Rafiei1, Eftekhari Hojat1, Arash Daryakar2, Leila Yusefkhani1, Behnam Rafiee3
1Department of Dermatology, Skin Research Center, Guilan University of Medical Sciences, Razi Hospital, Sardare Jangal St. Rasht, Iran; 2Department of Pathology, Skin Research Center, Guilan University of Medical Sciences, Razi Hospital, Sardare Jangal St. Rasht, Iran; 3epartment of Internal Medicine, University of Texas, MD Anderson Cancer Center 1515 Holcombe blvd. B4.4512, Houston, USA.
Corresponding author: Rana Rafiei, MD., E-mail: rafieirana@yahoo.com
Submission: 04.03.2016; Acceptance: 25.05.2016
DOI: 10.7241/ourd.20171.14
ABSTRACT
Anetoderma describes a localized laxity of the skin due to dermal elastolysis. It can be either primary or secondary to an inflammatory dermatosis. Rarely secondary anetoderma has been associated with mastocytosis. We report a 16-year-old man, with multiple pouch like lesions (anetoderma) mostly on the face, neck and upper trunk which had positive Darier`s sign. Anetodermic lesions developed two years ago without preceding urticaria pigmentosa lesions. In skin biopsy perivascular mast cell and eosinophil infiltrations were observed.
Key words: Anetoderma; Mastocytosis; Darier’s sign
INTRODUCTION
Anetodermic lesions are localized areas of flaccid or herniated saclike skin with focal loss of dermal elastic tissue. This rare condition has been classified as primary or idiopathic anetoderma which flaccid lesions occur in normal skin and secondary anetoderma which is preceded by inflammatory, autoimmune, infectious or neoplastic processes. The most common sites of anetoderma are the trunk, thighs and upper arms [1]. Cutaneous mastocytosis is characterized by accumulation of mast cells in the skin with heterogeneous clinical presentations [2], which include maculopapular lesions (urticarial pigmentosa), erythrodermic and telangiectatic aspect. Rare and atypical presentations of mastocytosis has been reported in literatures consist of bullous, pseudoxanthomatous, congenital dermographism and anetodermic lesions [3,4]. Degranulating mast cells could be able to degraded dermal elastin [5] and induce anetoderma. Mild trauma results in erythema and urticaria around mastocytosis macule which named Darier`s sign which helps us as a clinical guide for detecting anetodermic mastocytosis [2].
CASE REPORT
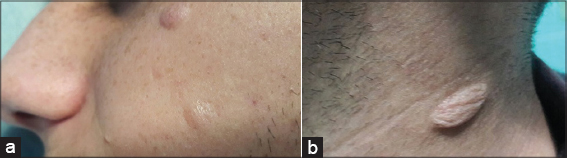
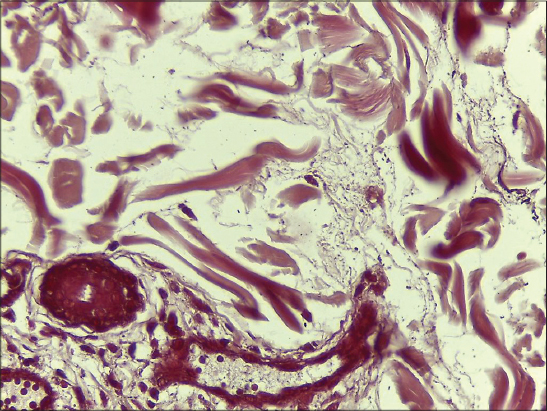
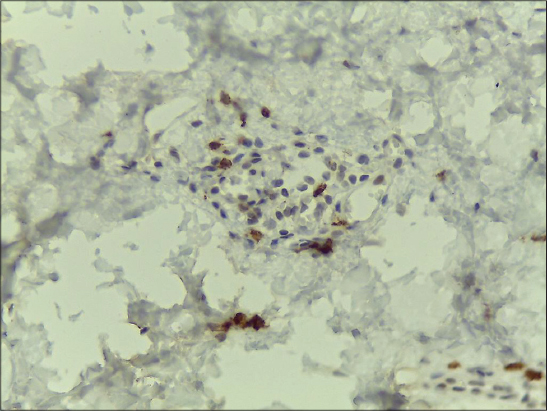
We present herein a 16-year-old man, with multiple asymptomatic flaccid pouch like lesions mostly on the face, neck and upper trunk. On skin examination the lesions could be depressed deeply without resistance (Figs. 1a and b). Stroking of the lesions resulted in erythema and swelling (positive Darier`s sign) but he had no dermographism on the normal skin. These lesions developed two years before the first visit without any inflammatory skin lesions such as acne vulgaris or insect bite. Past medical, drug and family history were negative. He had no atopy, allergic rhinitis or asthma. His parents did not remember any skin lesions during the infancy to suggest urticaria pigmentosa (UP). He mentioned that his lesions began as a soft skin color nodules which were gradually changed to flaccid pouch like lesions or spontaneously. Two skin biopsies were done. One from a soft nodule on the back and the other one from a pouch like lesion on the chin. Histopathologic sections of both specimens showed skin tissue covered by an atrophic epidermis under lied by edematous dermis with prominent separation of collagen bundles and perivascular infiltrations of lymphocytes, mast cells and eosinophils. Significant loss of elastic fibers in orcein staining was reported (Figs. 2a and b). No mucin identified in alcian blue stained sections. Immunohistochemistry (IHC) study revealed increased perivascular CD117 positive cells (Fig. 2c), which all of these findings were in favor of anetodermic mastocytosis.
Our patient had no systemic symptoms such as flushing, abdominal pain, diarrhea or bone pain. On clinical examination there was no organomegaly or lymphadenopathy. Laboratory evaluations including complete blood count, erythrocyte sedimentation rate, C- reactive protein, liver function test, urine analyses, were normal. Urinary 24-hour N-methyl histamine levels were slightly elevated. Due to lack of the systemic symptoms, serum tryptase level and bone marrow aspiration were not done. He complained of development of new lesions almost every six months, so phototherapy was considered for him.
Prior to the study, patient gave written consent to the examination and biopsy after having been informed about the procedure.
DISCUSSION
Anetoderma is a benign condition with focal loss of dermal elastic tissue resulting in localized areas of flaccid or herniated saclike skin, whose etiopathogenesis has been unkown. This rare disorder occurs mainly in women aged 20–40 years. In the most usual form, crops of round or oval, pink macules 0.5–1 centimeter in diameter develop on trunk, thighs and upper arms, less commonly on the neck and face and rarely elsewhere. The lesions remain unchanged throughout life, and new lesions often continue to develop for many years [6]. Anetoderma has been classified as primary and secondary. In primary anetoderma, patients do not remember prior inflammation in the lesions and no abnormal inflammatory infiltrate in dermis or microthrombosis are seen in skin biopsy. Secondary anetoderma develops after many different dermatosis including acne, varicella, syphilis, leprosy, sarcoidosis, mastocytosis, granuloma annulare, xanthoma, pilomatrichoma, lymphoma and lupus profundus [7].
Our case was interesting to us because secondary anetoderma associated with mastocytosis has been rarely reported in the literature. It has been observed in only a few patients with UP which is the most common type of mastocytosis in children [3]. Kalogeromitros, et al reported anetodermic lesions developed on the sites of UP lesions [8] but our patient`s family did not report any preceding UP lesions during the childhood and anetodermic lesions superimposed over the soft asymptomatic new skin color nodules or sometimes developed spontaneously. Positive Darier`s sign on anetodermic lesions was the only point for diagnosis which is pathognomonic for mastocytosis. So Darier`s sign should be investigated in anetodermic lesions with unknown origin in order to exclude cutaneous mastocytosis [2]. The exact mechanism of mechanical degranulation of tissue mast cells after rubbing the lesions is still obscure. Darier`s sign might reflect abnormal mechanosensitivity of mast cells in certain skin lesions [9].
Pathogenesis of anetoderma is unclear but degeneration of the elastic fibers could be mediated by close contact of degranulating mast cells with elastic fibers [5]. The role of elastase and other serine proteinases such as tryptase and chymase in mast cells has been postulated in anetoderma [10]. On the other hand elastin fragments are chemoattractant for phagocytes such as macrophage [5] which aggravate dermal elastolysis and result in anetoderma.
Our patients had no pruritus or UP lesions but he complained of new onset lesions during last two years. Del Pozo, et al reported phototherapy with ultra violet A and psoralen (PUVA) was effective in control of anetodermic mastocytosis [2]. So we scheduled phototherapy for him to prevent new lesions and he was followed for detecting any systemic symptoms of the mastocytosis in futures.
Abbreviations
(UP): Urticaria pigmentosa
(IHC): Immunohistochemistry
(PUVA): Psoralen- Ultraviolet A
Consent
The examination of the patient was conducted according to the Declaration of Helsinki principles.
REFERENCES
1. Aghaei S, Sodaifi M, Aslani FS, Mazharinia N, An unusual presentation of anetoderma:a case reportBMC Dermatol 2004; 4: 9-
2. Del Pozo J, Pimentel MT, Paradela S, Almagro M, Martinez W, Fonseca E, Anetodermic mastocytosis:response to PUVA therapyJ Dermatolog Treat 2007; 18: 184-7.
3. Fernandes IC, Teixeira Mdos A, Freitas I, Selores M, Alves R, Lima M, Adult mastocytosis:a review of the Santo Antonio Hospital ’s experience and an evaluation of World Health Organization criteria for the diagnosis of systemic diseaseAn Bras Dermatol 2014; 89: 59-66.
4. Rajesh J, Dogra S, Verma S, Mohanty SK, Handa S, Diffuse cutaneous mastocytosis:pseudoxanthomatous variantJ Dermatol 2002; 29: 354-6.
5. Sheu HM, Yu HS, Chang CH, Mast cell degranulation and elastolysis in the early stage of striae distensaeJ Cutan Pathol 1991; 18: 410-6.
6. Venencie PY, Winkelmann RK, Moore BA, Anetoderma:clinical findings, associations, and long term follow-up evaluationsArch Dermatol 1984; 120: 1032-9.
7. Sparsa A, Piette JC, Wechsler B, Amoura Z, Frances C, Anetoderma and its prothrombotic abnormalitiesJ Am Acad Dermatol 2003; 49: 1008-12.
8. Kalogeromitros D, Gregoriou S, Makris M, Georgala S, Kempuraj D, Theoharides TC, Secondary anetoderma associated with mastocytosisInt Arch Allergy Immunol 2007; 142: 86-8.
9. Kiorpelidou D, Gaitanis G, Bassukas ID, Darier’s sign:a model for studying dermographismIndian J Dermatol Venereol Leprol 2008; 74: 271-2.
10. Harvima IT, Naukkarinen A, Harvima RJ, Fraki JE, Immunoperoxidase and enzyme-histochemical demonstration of human skin tryptase in cutaneous mast cells in normal and mastocytoma skinArch Dermatol Res 1988; 280: 363-70.
Notes
Source of Support: Nil
Conflict of Interest: None declared.

Comments are closed.