Our Dermatol Online. 13; 4(2): 157-160
DOI:. 10.7241/ourd.20132.36
Date of submission: 03.01.2012 / acceptance: 13.02.2013
Conflicts of interest: None
A STUDY ON TINEA CAPITIS IN THE PRE SCHOOL AND SCHOOL GOING CHILDREN
Neerja Puri, Asha Puri
Department of Dermatology and Venereology, Punjab Health Systems Corporation, Ferozepur, Punjab, India
Corresponding author: Dr. Neerja Puri e-mail: neerjaashu@rediffmail.com
How to cite an article: Puri N, Puri A. A study on tinea capitis in the pre school and school going children. Our Dermatol Online. 2013; 4(2): 157-160.
Abstract
Introduction: Tinea capitis is a superficial fungal infection of scalp and hair caused by various species of dermatophytes. The incidence of Tinea capitis varies from country to country and region to region.
Material and Methods: tract Fifty patients from the preschool going population were selected for the study.
Results and Discussion: Clinical presentation of disease revealed that black dot to be the commonest (32%) followed by grey patch (28%), kerion (20%) and favus type was the least (1%). Direct microscopy of hair in KOH preparations revealed that all clinically suspected patients of Tinea capitis had endothrix type in 56% of cases and ectothrix type in 44%.
Key words: Tinea capitis; fungal; hair; ectothrix; endothrix
Introduction
Tinea capitis (TC) is a fungal infection of the scalp, hair follicles and hair shafts, especially common in the pediatric population and under tropical conditions [1,2]. The highest incidence is seen in children 3-7 years of age. The presence of symptoms like hyperkeratosis of scalp, seborrhea-like symptoms, excoriation secondary to pruritus, alopecia, broken hair or „black dot” appearance, cervical lymphadenopathy, pustules, or indurated or boggy plaques in a child should alert the dermatologist toward the possibility of TC [3,4]. TC is caused by various dermatophytes. The prevalence of various causative fungi varies according to the geographical area being studied [5,6]. TC can present as noninflammatory or inflammatory morphological variants. An early diagnosis is important to prevent transmission between children, especially siblings, and also to avoid possible scarring and permanent hair loss. The presence of tinea corporis or an id reaction in a child should also prompt a search for TC. As is true for most infectious diseases, the epidemiology of TC is in a constant state of flux, and varies considerably with respect to geography and specific population groups [7]. This fact prompted the present study, aimed at evaluating the clinical and etiologic profile of TC.
Aims
The major aim of this study was to determine the incidence, clinical presentation, age and sex distribution and seasonal variation of Tinea capitis among school going children. One objective was to relate the aetiological agent to the type of lesions.
Material and Methods
Fifty patients from the preschool going population were selected for the study from the dermatology outdoor department.There were 35 male children and 15 female children between 3 to 10 years of age. A detailed history was taken regarding the duration and pattern of hair loss. Demographic and socioeconomic data of the patients were recorded. Socioeconomic status of the children included patients in low income group, middle income group and high income group. Factors predisposing toward the spread of TC were analyzed. This included an assessment of the living conditions, history of any other family members affected and the hair care practices being followed. The patients were specifically asked about pets or any other prolonged contact with animals.Examination of the whole scalp was carried out to assess the type and extent of hair loss. The children included in our study were those presenting with patchy hair loss and easy pluckability of hair, with or without any associated inflammatory changes. Patients on any oral or topical antifungal therapy for the past 6 weeks were excluded from the study. Patients were classified according to the morphological types of TC as non inflammatory black dot (BD), gray patch (GP), inflammatory (pustular, kerion or favus) or mixed infection (any combination of the above). The patients were thoroughly examined to assess for any evidence of tinea corporis, nail involvement, id reaction or lymphadenopathy. Siblings of affected children as well as close family contacts were also examined to assess for any hair loss. The patients were asked to come after head wash so as to remove any oil from the scalp. For all the patients, skin scrapings and hair fragments were collected from the affected areas in an aseptic manner. The material collected on a slide was immersed in potassium hydroxide (10% KOH) to prepare smears for microscopic examination. Both macroscopical and microscopical examinations were performed to identify the dermatophyte isolates. The slides were assessed under a low-power microscope to look for fungal arthrospores or any hyphae. If the spores were located on the surface of the hair shaft, without causing any distortion of hair architecture, the infection was classified as ectothrix. If the spores were seen inside the shaft and were destroying the hair fragment architecture, the infection was classified as endothrix. Presence of both the patterns in the same specimen was also recorded. The samples were also inoculated on Sabouraud’s Dextrose Agar; with and without antibiotics (chloramphenicol, gentamicin and cycloheximide). This was done to identify the causative species involved. The clinical, microbiological and etiologic data were collected and correlated.
Results
The data was collected and the results were analysed.
| Sr no | Morphological pattern | Number | Percentage |
| 1 | Black dot type | 16 | 32 |
| 2 | Grey patch | 14 | 28 |
| 3 | Kerion | 10 | 20 |
| 4 | Pustular | 6 | 12 |
| 5 | Mixed | 4 | 8 |
| TOTAL | 50 | 100 |
Table I. Morphological pattern of patients
| Sr no | Clinical type | Endothrix (28) | Ectothrix (22) |
| 1 | Black dot type (n=16) | 10 (62.5%) | 6 (37.5%) |
| 2 | Grey patch type (n=14) | 6 (42.8%) | 8 (57.1%) |
| 3 | Kerion (n = 10) | 6 (60%) | 4 (40%) |
| 4 | Pustular type (n=6) | 4 (66.6%) | 2 (33.3%) |
| 5 | Mixed (n=4) | 2 (33.3%) | 2 (33.3%) |
Table II. Clinical and microscopic types of tinea capitis
| Sr no | Species | Black dot | Grey patch | Kerion | Pustular | Mixed |
| 1 | Trichophyton violaceum | 14 | 10 | 6 | 4 | 3 |
| 2 | Trichophyton rubrum | – | 1 | – | – | – |
| 3 | Trichophyton tonsurans | – | – | 2 | – | – |
| 4 | Microsporum audounii | – | 2 | – | – | 1 |
| 5 | No growth | 2 | 4 | 2 | 2 | – |
Table III. Causative agent of tinea capitis
Discussion
It was seen that the majority of children were in the age group of 3-10 years.Beyond this age group, the incidence declines because of the onset of puberty and seborrhea. Regarding distribution, male children patients numbered female children and male : female ratio was 2:1. Various conflicting views exist regarding the sexual predominance of TC. The low frequency in the females could be due to custom of regular application of vegetable oil over the scalp which has fungistatic properties. Some authorities believe that TC may be common in boys due to shorter hair, allowing easy access for circulating spores, while others believe that it may be more common in girls due to tight hair braiding. 60% patients showed inflammatory (Fig. 1) tinea capitis and 32% patients showed non inflammatory tinea capitis. Family history of TC which was seen in 29% of patients may be due to sharing of articles like towels, combs, cloth cap etc. by other family members. Fungi was recovered on culture in 65% patients and regarding the causative agent of fungi no growth was seen in 20% patients. 82% of the children belonged to the lower middle and 22% belonged to the lower income groups. There were 15 sets of siblings among these patients, with each set having two to three children. A history of sharing of combs and hair accessories was elicited in 68% of the patients. Only 15% gave a history of pets at home or prolonged contact with animals. An attempt was made to correlate the clinical and microscopic types of TC. It was seen that although the endothrix pattern was more common for BDTC, the ectothrix pattern is also seen in a large number of cases. Similarly, ectothrix invasion is more common for gray patch tinea capitis (GPTC), although endothrix cases can also be seen. Also, most of the cases with a mixed pattern of invasion on KOH belonged to the GP + BD morphology. Culture specimens from all the cases were examined. No growth at the end of 6 weeks was recorded in 20% of the cases (10 cases). Of those showing growth of fungal elements, Trichophyton violaceum was the most common isolate in 74% (37 cases). This was followed by T. rubrum (one case), T. tonsurans (two cases), Microsporum audouinii (two cases). A correlation of fungal species isolated with the clinical type of TC was drawn out. It can be seen that Trichophyton violaceum was responsible for BDTC (Fig. 2) in most of the cases. M. audouinii were responsible for a GPTC pattern and T. tonsurans was isolated from cases of kerion (Fig. 3). Majority of TC cases were from urban area (78%) and family history of dermatophytoses was present in 25% of cases. There was seasonal incidence of cases of TC. Incidence of disease was slightly higher (45%) in post monsoon period (July-October) and the percentage of cases from January to April was also 38%. Incidence was low, 5% and 3.5% in extreme summer (May-June) and begining of winter (Novermber-December) respectively. Clinical presentation of disease revealed that black dot to be the commonest (32%) followed by grey patch (28%), kerion (20%) and favus type was the least (1%). Direct microscopy of hair in KOH preparations revealed that all clinically suspected patients of TC had endothrix type in 56% of cases and ectothrix type in 44%. The percentage of endothrix infection in black dot type was 62.5%, grey patch 42.8%, Kerion 60% and favus 1%, whereas ectothrix hair involvement was seen only in grey patch (57.2%) and kerion (40%). It was observed that T. violaceum was main isolate from black dot type while T. mentagrophyte was the main isolate from grey patch and kerion lesions. TC is a common fungal infection, particularly among children in urban regions [8-10]. More often than not, it presents with mild scaling and little hair loss, which is reversible. However, in a few cases, it may be characterized by intense inflammation and subsequent cicatricial alopecia, which causes permanent cosmetic disfigurement. Also, the infection is highly contagious and, hence, needs to be recognized and treated early to prevent transmission to siblings and costudents [11-13]. Awareness in patients regarding disease was good (88%) and also incidence was higher in urban areas compared to rural population.
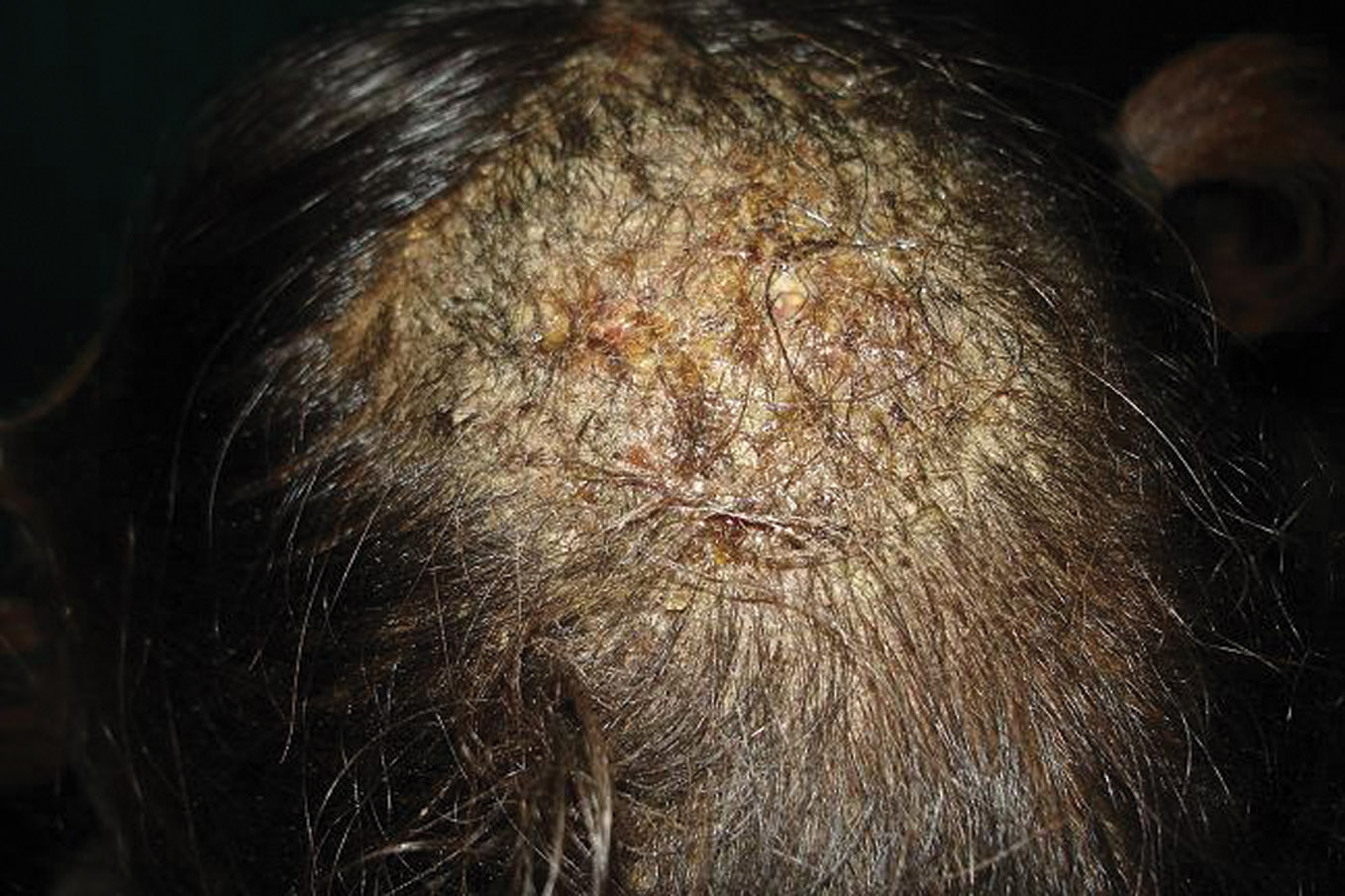 Figure 1. Inflammatory tinea capitis in a 6 year old child
|
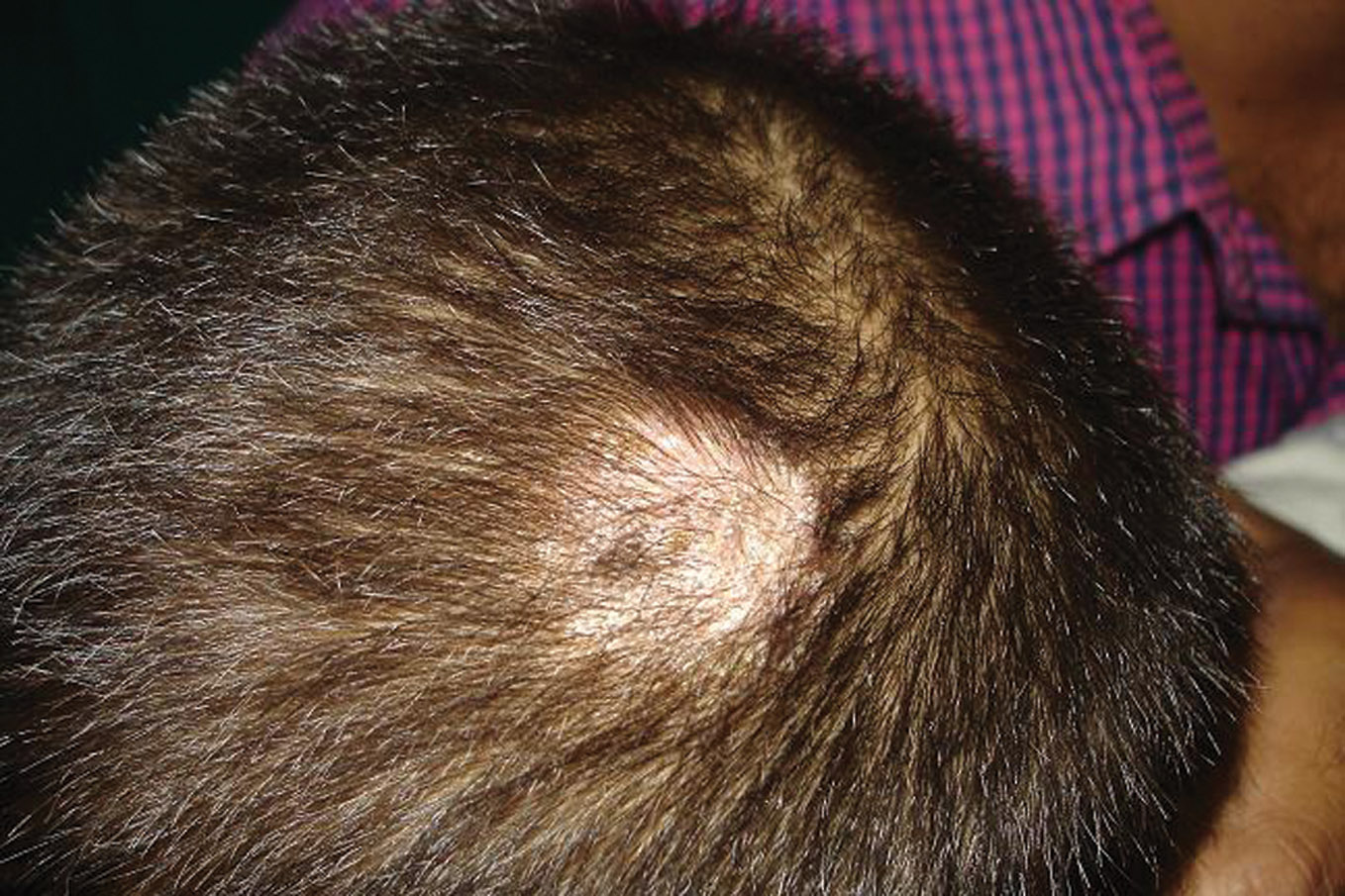 Figure 2. Black dot tinea capitis in a 4 year old male child
|
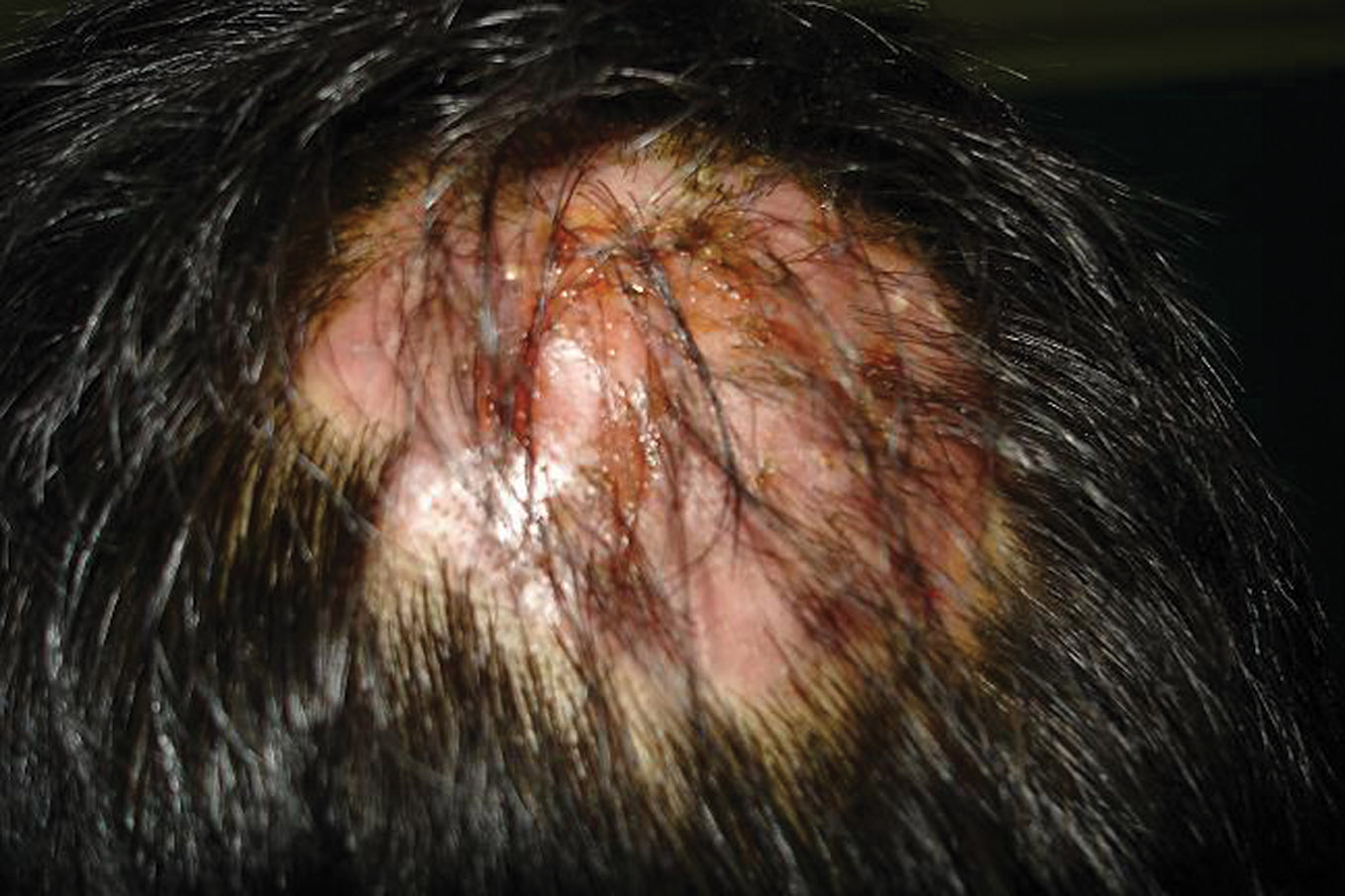 Figure 3. Kerion in a 3 year old child
|
Conclusion
Our study threw up interesting findings. BDTC was the most common type of TC and Trichophyton violaceum was the most common species isolated. KOH examination and culture were useful diagnostic methods. However, the clinical morphology or KOH findings were not found to be clearly predictive of the species involved. Mixed patterns were observed both on clinical examination as well as on KOH examination. In our study results of microscopic examination of hair in KOH mount when correlated with culture, it was seen that majority of dermatophytes isolated was Trichophyton violaceum which caused endothrix type of hair infection. Also, the percentage of dermatophytes on culture in our study was 65%.
REFERENCES
1. Elewski BE: Tinea capitis: A current perspective. J Am Acad Dermatol. 2000;42:1-20. 2. Alvarez MS, Silverberg NB: Tinea capitis. Cutis. 2006;78:189-96. 3. Chen BK, Friedlander SF: Tinea capitis update: a continuing conflict with an old adversary. Curr Opin Pediatr. 2001;13:331-5. 4. Trovato MJ, Schwartz AR, Janninger CK: Tinea capitis: current concepts in clinical practice. Cutis. 2006;77:93-9. 5. Jha BN, Garg VK, Agrawal S, Khanal B, Agarwalla A: Tinea capitis in Eastern Nepal. Int J Dermatol. 2006;45:100-2. 6. Kalla G, Begra B, Solanki A, Goyal A, Batra A: Clinico mycological study of Tinea capitis in Desert District of Rajasthan. Ind J Dermatol Venereol Leprol. 1995;61:342-5. 7. Jahangir M, Hussain I, Khurshid K, Haroon TS: A clinico-etiologic correlation in Tinea capitis. Int J Dermatol. 1999;38:275-8. 8. Sidat MM, Correia D, Buene TP: Tinea capitis among children at one suburban primary school in the city of Maputo, Mozambique. Revista da Sociedade de Medicina Tropical. 2000;40:43-4-5. 9. Mills CM, Philpot CM: Tinea capitis in south Wales-observations in change of causative fungi. Clin Exp Dermatol. 1994;19:473-5. 10. Jain N, Sharma M, Saxena VN: Clinico-mycological profile of dermatophytosis in Jaipur, Rajasthan. Indian J Dermatol Venereol Leprol. 2008;74:274-5. 11. Kumar AG, Lakshmi N: Tinea capitis in Tirupati. Ind J Pathol Microbiol. 1990;33:360-3. 12. Singal A, Rawat S, Bhattacharya S, Mohanty S, Baruah MC: Clinico- mycological profile of tinea capitis in North India and response to griseofulvin. J Dermatol. 2001;28:22-6. 13. Hussain I, Aman S, Haroon TS, Jahangir M, Nagi AH: Tinea capitis in Lahore, Pakistan. Int J Dermatol. 1994;33:255-7.
Comments are closed.